A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
Correct Answer :
B. Two constant volume
Related Questions
Which of the following is an irreversible cycle?
Carnot
Stirling
Ericsson
None of the above
Which of the following is the correct statement of the second law of thermodynamics?
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work.
It is impossible to transfer heat from a body at a lower temperature to a higher temperature, without the aid of an external source.
There is a definite amount of mechanical energy, which can be obtained from a given quantity of heat energy.
All of the above
In the below figure, the plastic range occurs
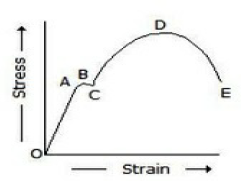
Before point A
Beyond point A
Between points A and D
Between points D and E
During which of the following process does heat rejection takes place in Carnot cycle?
Isothermal expansion
Isentropic expansion
Isothermal compression
Isentropic compression
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
Strain energy is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of a specimen
Maximum strain energy which can be stored in a body
Proof resilience per unit volume of a material
The calorific value of gaseous fuel is expressed in
kJ
kJ/kg
kJ/m2
kJ/m3
For a perfect gas, according to Boyle's law (where p = Absolute pressure, v = Volume, and T = Absolute temperature)
p v = constant, if T is kept constant
v/T = constant, if p is kept constant
p/T = constant, if v is kept constant
T/p = constant, if v is kept constant
Resilience is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of the specimen maximum strain
Energy which can be stored in a body
None of the above
If a part is constrained to move and heated, it will develop
Principal stress
Tensile stress
Compressive stress
Shear stress
The section modulus of a circular section about an axis through its C.G., is
πd²/4
πd²/16
πd3/16
πd3/32
The deformation per unit length is called
Tensile stress
Compressive stress
Shear stress
Strain
The ratio of elongation in a prismatic bar due to its own weight (W) as compared to another similar bar carrying an additional weight (W) will be
1 : 2
1 : 3
1 : 4
1 : 2.5
A column of length (l) with both ends fixed may be considered as equivalent to a column of length __________ with both ends hinged.
l/8
l/4
l/2
l
The extremeties of any diameter on Mohr's circle represent
Principal stresses
Normal stresses on planes at 45°
Shear stresses on planes at 45°
Normal and shear stresses on a plane
The percentage reduction in area of a cast iron specimen during tensile test would be of the order of
More than 50 %
25-50 %
10-25 %
Negligible
In an extensive property of a thermodynamic system
Extensive heat is transferred
Extensive work is done
Extensive energy is utilised
None of these
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
According to Kelvin-Planck's statement of second law of thermodynamics,
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is possible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is impossible to construct a device which operates in a cyclic process and produces no effect other than the transfer of heat from a cold body to a hot body
None of the above
The buckling load for a given material depends on
Slenderness ratio and area of cross-section
Poisson's ratio and modulus of elasticity
Slenderness ratio and modulus of elasticity
Slenderness ratio, area of cross-section and modulus of elasticity
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
The weakest section of a diamond riveting is the section which passes through
The first row
The second row
The central row
One rivet hole of the end row
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
In order to know whether a column is long or short, we must know its
Ultimate shear stress of the column
Factor of safety
Torque resisting capacity
Slenderness ratio
Which of the following statement is incorrect?
The liquid fuels consist of hydrocarbons.
The liquid fuels have higher calorific value than solid fuels.
The solid fuels have higher calorific value than liquid fuels.
A good fuel should have low ignition point.
Carnot cycle has maximum efficiency for
Petrol engine
Diesel engine
Reversible engine
Irreversible engine
One reversible heat engine operates between 1600 K and T2 K and another reversible heat engine operates between T2 K and 400 K. If both the engines have the same heat input and output, then temperature T2 is equal to
800 K
1000 K
1200 K
1400 K
The amount of heat generated per kg of fuel is known as
Calorific value
Heat energy
Lower calorific value
Higher calorific value
A cycle consisting of two constant pressure and two isentropic processes is known as
Carnot cycle
Stirling cycle
Otto cycle
None of these
