According to Avogadro's law
The product of the gas constant and the molecular mass of an ideal gas is constant
The sum of partial pressure of the mixture of two gases is sum of the two
Equal volumes of all gases, at the same temperature and pressure, contain equal number of molecules
All of the above
Correct Answer :
C. Equal volumes of all gases, at the same temperature and pressure, contain equal number of molecules
Related Questions
The efficiency and work ratio of a simple gas turbine cycle are
Low
Very low
High
Very high
Tensile strength of a material is obtained by dividing the maximum load during the test by the
Area at the time of fracture
Original cross-sectional area
Average of (A) and (B)
Minimum area after fracture
The torsional rigidity of a shaft is expressed by the
Maximum torque it can transmit
Number of cycles it undergoes before failure
Elastic limit up to which it resists torsion, shear and bending stresses
Torque required to produce a twist of one radian per unit length of shaft
Select the correct statement as per Charles' law
p.v = constant, if T is kept constant
v/T = constant, if p is kept constant
p/T = constant, if v is kept constant
T/p = constant, if v is kept constant
Otto cycle consists of
Two constant volume and two isentropic processes
Two constant pressure and two isentropic processes
Two constant volume and two isothermal processes
One constant pressure, one constant volume and two isentropic processes
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
A circular shaft fixed at, A has diameter D for half of its length and diameter D/2 over the other half, as shown in the below figure. If the rotation of B relative to A is 0.1 radian, the rotation of C relative to B will be
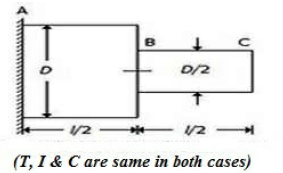
0.4 radian
0.8 radian
1.6 radian
3.2 radian
When two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This statement is called
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kelvin Planck's law
Shear modulus is the ratio of
Linear stress to linear strain
Linear stress to lateral strain
Volumetric strain to linear strain
Shear stress to shear strain
The point of contraflexure is a point where
Shear force changes sign
Bending moment changes sign
Shear force is maximum
Bending moment is maximum
Carnot cycle consists of
Two constant volume and two isentropic processes
Two isothermal and two isentropic processes
Two constant pressure and two isentropic processes
One constant volume, one constant pressure and two isentropic processes
One kg of carbon requires 4/3 kg of oxygen and produces __________ kg of carbon monoxide gas.
8/3
11/3
11/7
7/3
If a material expands freely due to heating it will develop
Thermal stresses
Tensile stress
Bending
No stress
The neutral axis of the cross-section a beam is that axis at which the bending stress is
Zero
Minimum
Maximum
Infinity
The property of a working substance which increases or decreases as the heat is supplied or removed in a reversible manner, is known as
Enthalpy
Internal energy
Entropy
External energy
The sum of internal energy (U) and the product of pressure and volume (p.v) is known as
Workdone
Entropy
Enthalpy
None of these
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
Which of the following is correct?
Gauge pressure = Absolute pressure + Atmospheric pressure
Absolute pressure = Gauge pressure + Atmospheric pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
The __________ states that change of internal energy of a perfect gas is directly proportional to the change of temperature.
Boyle's law
Charle's law
Gay-Lussac law
Joule's law
Flow stress corresponds to
Fluids in motion
Breaking point
Plastic deformation of solids
Rupture stress
The oxygen atom is ________ times heavier than the hydrogen atom.
2
8
16
32
In the below figure, the plastic range occurs
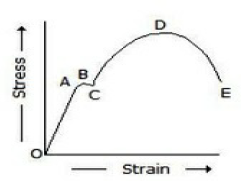
Before point A
Beyond point A
Between points A and D
Between points D and E
Two shafts 'A' and 'B' transmit the same power. The speed of shaft 'A' is 250 r.p.m. and that of shaft 'B' is 300 r.p.m.
The shaft 'B' has the greater diameter
The shaft 'A' has the greater diameter
Both are of same diameter
None of these
For a perfect gas, according to Boyle's law (where p = Absolute pressure, v = Volume, and T = Absolute temperature)
p v = constant, if T is kept constant
v/T = constant, if p is kept constant
p/T = constant, if v is kept constant
T/p = constant, if v is kept constant
Stirling and Ericsson cycles are
Reversible cycles
Irreversible cycles
Semi-reversible cycles
Quasi-static cycles
A column is said to be a short column, when
Its length is very small
Its cross-sectional area is small
The ratio of its length to the least radius of gyration is less than 80
The ratio of its length to the least radius of gyration is more than 80
In a belt drive, the pulley diameter is doubled, the belt tension and pulley width remaining same. The changes required in key will be
Increase key length
Increase key depth
Increase key width
Double all the dimensions
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
In the torsion equation T/J = τ/r = Gθ/ L, the term J/R is called
Shear modulus
Section modulus
Polar modulus
None of these
The unit of Young's modulus is
mm/mm
kg/cm
Kg
kg/cm²
