All perfect gases change in volume by 1/273th of its original volume at 0°C for every 1°C change in temperature, when the pressure remains constant. This statement is called
Boyle's law
Charles' law
Gay-Lussac law
Joule's law
Correct Answer :
B. Charles' law
Related Questions
All perfect gases change in volume by 1/273th of its original volume at 0°C for every 1°C change in temperature, when the pressure remains constant. This statement is called
Boyle's law
Charles' law
Gay-Lussac law
Joule's law
A body is subjected to a tensile stress of 1200 MPa on one plane and another tensile stress of 600 MPa on a plane at right angles to the former. It is also subjected to a shear stress of 400 MPa on the same planes. The maximum normal stress will be
400 MPa
500 MPa
900 MPa
1400 MPa
Hooke's law holds good up to
Yield point
Limit of proportionality
Breaking point
Elastic limit
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The extremeties of any diameter on Mohr's circle represent
Principal stresses
Normal stresses on planes at 45°
Shear stresses on planes at 45°
Normal and shear stresses on a plane
A thin mild steel wire is loaded by adding loads in equal increments till it breaks. The extensions noted with increasing loads will behave as under
Uniform throughout
Increase uniformly
First increase and then decrease
Increase uniformly first and then increase rapidly
If the rivets in adjacent rows are staggered and the outermost row has only one rivets, the arrangement of the rivets is called
Chain riveting
Zigzag riveting
Diamond riveting
Crisscross riveting
The __________ is obtained when carbonisation of coal is carried out at 500° to 700° C.
Soft coal
Hard coal
Pulverised coal
Bituminous coal
Reversed Joule cycle is known as
Carnot cycle
Bell-Coleman cycle
Rankine cycle
Stirling cycle
Stirling and Ericsson cycles are
Reversible cycles
Irreversible cycles
Semi-reversible cycles
Adiabatic irreversible cycles
Youngs modulus of a wire is defined as the stress which will increase the length of wire compared to its original length by
Half
Same amount
Double
One-fourth
Efficiency of a riveted joint is the ratio of its strength (max. load it can resist without failure) to the strength of the un-punched plate in
Tension
Compression
Bearing
Any one of the above
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then minimum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
The property of a material by virtue of which it can be beaten or rolled into plates is called
Malleability
Ductility
Plasticity
Elasticity
A thin cylindrical shell of diameter (D) and thickness (t) is subjected to an internal pressure (p). The ratio of longitudinal strain to volumetric strain is
(m - 1)/ (2m - 1)
(2m - 1)/ (m - 1)
(m - 2)/ (3m - 4)
(m - 2)/ (5m - 4)
The ratio of elongation in a prismatic bar due to its own weight (W) as compared to another similar bar carrying an additional weight (W) will be
1 : 2
1 : 3
1 : 4
1 : 2.5
The rivets are used for __________ fastenings.
Permanent
Temporary
Semi-permanent
None of these
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then maximum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
The ratio of lateral strain to the linear strain within elastic limit is known as
Young's modulus
Bulk modulus
Modulus of rigidity
Poisson's ratio
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
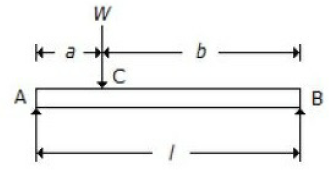
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
The atomic mass of sulphur is
12
14
16
32
The atomic mass of nitrogen is __________ oxygen.
Equal to
Less than
More than
None of these
The limit of eccentricity for no tensile conditions for a column of circular section of diameter (D) is
d/4
d/8
d/12
d/16
The smallest quantity of a substance, which can exist by itself in a chemically recognizable form is known as
Element
Compound
Atom
Molecule
Which is the false statement about true stress strain method?
It does not exist
It is more sensitive to changes in both metallurgical and mechanical conditions
It gives a more accurate picture of the ductility
It can be correlated with stress strain values in other tests like torsion, impact, combined stress tests etc.
The oxygen atom is ________ times heavier than the hydrogen atom.
2
8
16
32
The columns whose slenderness ratio is less than 80, are known as
Short columns
Long columns
Weak columns
Medium columns
Percentage reduction of area in performing tensile test on cast iron may be of the order of
50 %
25 %
0 %
15 %
The absolute zero temperature is taken as
-273°C
73°C
237°C
-237°C
The expansion ratio (r) is the ratio of (where v1 = Volume at the beginning of expansion, and v2 = Volume at the end of expansion)
v1/v2
v2/v1
(v1 + v2)/v1
(v1 + v2)/v2
