An isothermal process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
Correct Answer :
A. Boyle's law
Related Questions
The atomic mass of sulphur is
12
14
16
32
A beam is loaded as cantilever. If the load at the end is increased, the failure will occur
In the middle
At the tip below the load
At the support
Anywhere
Which of the following statement is incorrect?
The liquid fuels have higher calorific value than solid fuels
The solid fuels have higher calorific value than liquid fuels
A good fuel should have low ignition point
The liquid fuels consist of hydrocarbons
If Th is the torque resisting capacity of a hollow shaft and Ts is that of a solid shaft, of the same material, length and weight. Then,
Th > Ts
Th < Ts
Th = Ts
None of these
The deformation of a bar under its own weight compared to the deformation of same body subjected to a direct load equal to weight of the body is
Same
Double
Half
Four times
One kg of carbon monoxide requires _______kg of oxygen to produce 11/7 kg of carbon dioxide gas.
11/7
9/7
4/7
All of the above
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
A steel bar of 5 mm is heated from 25°C to 45°C and it is free to expand. The bar will induce
No stress
Shear stress
Tensile stress
Compressive stress
The maximum diameter of the hole that can be punched from a plate of maximum shear stress 1/4th of its maximum crushing stress of punch, is equal to (where t = Thickness of the plate)
t
2t
4t
8t
Diamond riveted joint can be adopted in the case of following type of joint
Butt joint
Lap joint
Double riveted lap joints
All types of joints
When a bar is cooled to - 5°C, it will develop
No stress
Shear stress
Tensile stress
Compressive stress
Steam coal is a
Pulverised coal
Brown coal
Coking bituminous coal
Non-coking bituminous coal
When a body is subjected to a direct tensile stress (σx) in one plane accompanied by a simple shear stress (τxy), the maximum shear stress is
(σx/2) + (1/2) × √(σx² + 4 τ²xy)
(σx/2) - (1/2) × √(σx² + 4 τ²xy)
(σx/2) + (1/2) × √(σx² - 4 τ²xy)
(1/2) × √(σx² + 4 τ²xy)
The heat energy stored in the gas and used for raising the temperature of the gas is known as
External energy
Internal energy
Kinetic energy
Molecular energy
A tri-atomic molecule consists of __________ atoms.
One
Two
Three
Four
Reversed Joule cycle is known as
Carnot cycle
Bell-Coleman cycle
Rankine cycle
Stirling cycle
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
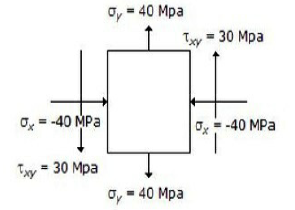
10 MPa
30 MPa
50 MPa
100 MPa
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
Reversed joule cycle is called
Carnot cycle
Rankine cycle
Brayton cycle
Bell Coleman cycle
Principal planes are planes having
Maximum shear stress
No shear stress
Minimum shear stress
None of the above
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
Carnot cycle consists of
Two constant volume and two isentropic processes
Two isothermal and two isentropic processes
Two constant pressure and two isentropic processes
One constant volume, one constant pressure and two isentropic processes
Which of the following cycles has maximum efficiency?
Rankine
Stirling
Carnot
Brayton
The ideal efficiency of a Brayton cycle without regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
The following cycle is used for air craft refrigeration
Brayton cycle
Joule cycle
Carnot cycle
Reversed Brayton cycle
The most probable velocity of the gas molecules is given by
√(KT/m)
√(2KT/m)
√(3KT/m)
√(5KT/m)
The expansion ratio (r) is the ratio of (where v1 = Volume at the beginning of expansion, and v2 = Volume at the end of expansion)
v1/v2
v2/v1
(v1 + v2)/v1
(v1 + v2)/v2
Kerosene is distilled at
65° to 220°C
220° to 345°C
345° to 470°C
470° to 550°C
The gas turbine cycle with regenerator improves
Thermal efficiency
Work ratio
Avoids pollution
None of these
When shear force at a point is zero, then bending moment is _________ at that point.
Zero
Minimum
Maximum
Infinity
