As the temperature is lowered towards the absolute zero, the value of ∂(ΔF)/∂T, then approaches
Unity
Zero
That of the heat of reaction
Infinity
Correct Answer :
B. Zero
Related Questions
The ammonia synthesis reaction represented by N2 + 3H2 2NH3; ΔH = - 22.4 kcal, is
Endothermic
Exothermic
Isothermal
Adiabatic
After throttling, gas temperature
Decreases
Increases
Remain same
May increase or decrease; depends on the nature of the gas
Filling of gas from a high pressure cylinder into small bottles is an example of a/an __________ process.
Equilibrium
Adiabatic
Steady
Unsteady
Trouton's ratio of __________ liquids is calculated using Kistyakowsky equation.
Polar
Non-polar
Both (A) & (B)
Neither (A) nor (B)
Efficiency of a heat engine working on Carnot cycle between two temperature levels depends upon the
Two temperatures only
Pressure of working fluid
Mass of the working fluid
Mass and pressure both of the working fluid
Which of the following decreases with increase in pressure?
Melting point of ice
Melting point of wax
Boiling point of liquids
None of these
__________ decreases during adiabatic throttling of a perfect gas.
Entropy
Temperature
Enthalpy
Pressure
The value of gas constant 'R' is
1.987 cal/gm mole °K
1.987 BTU/lb. mole °R
Both (A) and (B)
Neither (A) nor (B)
Entropy, which is a measure of the disorder of a system, is:
Independent of pressure
Independent of temperature
Zero at absolute zero temperature for a perfect crystalline substance
All (A), (B) & (C)
Equation which relates pressure, volume and temperature of a gas is called the
Equation of state
Gibbs Duhem equation
Ideal gas equation
None of these
For water at 300°C, it has a vapour pressure 8592.7 kPa and fugacity 6738.9 kPa Under these conditions, one mole of water in liquid phase has a volume of 25.28 cm3 and that in vapour phase in 391.1 cm3.Fugacity of water (in kPa) at 9000 kPa will be
6738.9
6753.5
7058.3
9000
What happens in a reversible adiabatic expansion process?
Heating takes place
Cooling takes place
Pressure is constant
Temperature is constant
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
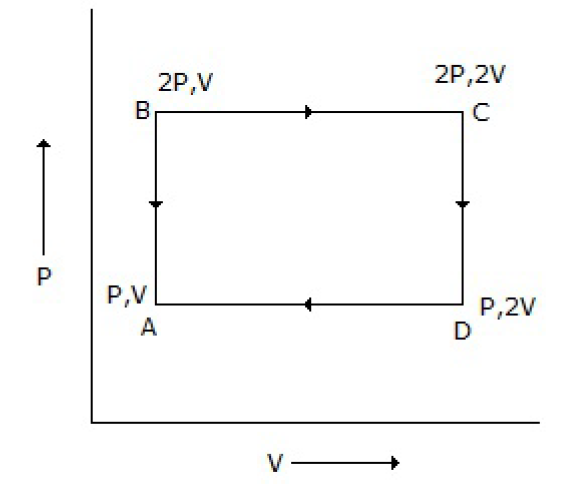
PV
2PV
PV/2
0
High pressure steam is expanded adiabatically and reversibly through a well insulated turbine, which produces some shaft work. If the enthalpy change and entropy change across the turbine are represented by ΔH and ΔS respectively for this process:
Δ H = 0 and ΔS = 0
Δ H ≠ 0 and ΔS = 0
Δ H ≠ 0 and ΔS ≠ 0
Δ H = 0 and ΔS ≠ 0
In the ammonia synthesis reaction, N2 + 3H2 2NH3 + 22.4 kcal, the formation of NH3 will be favoured by
High temperature
Low pressure
Low temperature only
Both low temperature and high pressure
Domestic refrigerator usually works on the __________ refrigeration cycle.
Carnot
Air
Absorption
vapour-ejection
Steam undergoes isentropic expansion in a turbine from 5000 kPa and 400°C (entropy = 6.65 kJ/kg K) to 150 kPa) (entropy of saturated liquid = 1.4336 kJ/kg. K, entropy of saturated vapour = 7.2234 kJ/kg. K) The exit condition of steam is
Superheated vapour
Partially condensed vapour with quality of 0.9
Saturated vapour
Partially condensed vapour with quality of 0.1
In polytropic process (PVn = constant), if n = 1; it means a/an __________ process.
Adiabatic
Reversible
Isothermal
None of these
Pick out the wrong statement.
Trouton's ratio of non-polar liquids is calculated using Kistyakowsky equation
Thermal efficiency of a Carnot engine is always less than 1
An equation relating pressure, volume and temperature of a gas is called ideal gas equation
None of these
dW and dq are not the exact differential, because q and W are
State functions
Path functions
Intensive properties
Extensive properties
Molar heat capacity of water in equilibrium with ice at constant pressure is __________ Kcal/kg mole. °K
0
∞
50
100
The third law of thermodynamics states that the
Heat capacity of a crystalline solid is zero at absolute zero temperature
Heat transfer from low temperature to high temperature source is not possible without external work
Gases having same reduced properties behaves similarly
None of these
Which is a state function?
Specific volume
Work
Pressure
Temperature
When a gas is expanded from high pressure region to low pressure region; temperature change occurs. This phenomenon is related to the
Gibbs-Duhem equation
Gibbs-Helmholtz equation
Third law of thermodynamics
Joule-Thomson effect
Solid and liquid phases of a substance are in equilibrium at the
Critical temperature
Melting point
Freezing point
Both (B) and (C)
The free energy change for a chemical reaction is given by (where, K = equilibrium constant)
RT ln K
-RT ln K
-R ln K
T ln K
Which of the following identities can be most easily used to verify steam table data for superheated steam?
(∂T/∂V)S = (∂p/∂S)V
(∂T/∂P)S = (∂V/∂S)P
(∂P/∂T)V = (∂S/∂V)T
(∂V/∂T)P = -(∂S/∂P)T
(∂T/∂P)H is the mathematical expression for
Specific heat at constant pressure (Cp)
Specific heat at constant volume (Cv)
Joule-Thompson co-efficient
None of these
Joule-Thomson experiment is
Isobaric
Adiabatic
Isenthalpic
Both (B) & (C)
A Carnot cycle consists of the following steps:
Two isothermal and two isentropic
Two isobaric and two isothermal
Two isochoric and two isobaric
Two isothermals and two isochoric
