Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
Correct Answer :
C. When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
Related Questions
One kilowatt is equal to
1 N-m/s
100 N-m
1000 N-m/s
1 × 106 N-m/s
The ratio of maximum shear stress developed in a rectangular beam and a circular beam of the same cross-sectional area is
2/3
3/4
1
9/8
A fletched beam is used to
Change the shape of the beam
Effect the saving in material
Equalise the strength in tension and compression
Increase the cross-section of the beam
When gas is heated at constant pressure, the heat supplied is utilised in
Increasing the internal energy of gas
Doing some external work
Increasing the internal energy of gas and also for doing some external work
None of the above
If the depth is kept constant for a beam of uniform strength, then its width will vary in proportional to
Bending moment (i.e. M)
Bending moment² (i.e. M²)
Bending moment³ (i.e. M³)
Bending moment⁴ (i.e. M⁴)
The calorific value of gaseous fuel is expressed in
kJ
kJ/kg
kJ/m2
kJ/m3
The relation between Young's modulus (E), shear modulus (C) and bulk modulus (K) is given by
E = 3K.C/(3K + C)
E = 6K.C/(3K + C)
E = 9K.C/(3K + C)
E = 12K.C/(3K + C)
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
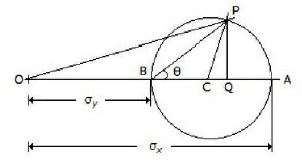
OC
OP
OQ
PQ
The thermodynamic difference between a Rankine cycle working with saturated steam and the Carnot cycle is that
Carnot cycle can't work with saturated steam
Heat is supplied to water at temperature below the maximum temperature of the cycle
A Rankine cycle receives heat at two places
Rankine cycle is hypothetical
The limit of eccentricity for no tensile conditions for a column of circular section of diameter (D) is
d/4
d/8
d/12
d/16
The ratio of specific heat at constant pressure (cp) and specific heat at constant volume (cv) is always __________ one.
Equal to
Less than
Greater than
None of these
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The neutral axis of the cross-section a beam is that axis at which the bending stress is
Zero
Minimum
Maximum
Infinity
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then maximum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
The property of a material by virtue of which it can be beaten or rolled into plates is called
Malleability
Ductility
Plasticity
Elasticity
The maximum diameter of the hole that can be punched from a plate of maximum shear stress 1/4th of its maximum crushing stress of punch, is equal to (where t = Thickness of the plate)
t
2t
4t
8t
The behaviour of a perfect gas, undergoing any change in the variables which control physical properties, is governed by
Boyle's law
Charles' law
Gay-Lussac law
All of these
The ultimate tensile stress of mild steel compared to ultimate compressive stress is
Same
More
Less
Unpredictable
An open system is one in which
Heat and work crosses the boundary of the system, but the mass of the working substance does not crosses the boundary of the system
Mass of the working substance crosses the boundary of the system but the heat and work does not crosses the boundary of the system
Both the heat and work as well as mass of the working substance crosses the boundary of the system
Neither the heat and work nor the mass of the working substance crosses the boundary of the system
A molecule consisting of one atom is known as
Mono-atomic
Di-atomic
Tri-atomic
Poly-atomic
The processes occuring in open system which permit the transfer of mass to and from the system, are known as
Flow processes
Non-flow processes
Adiabatic processes
None of these
The value of specific heat at constant pressure (cp) is __________ that of at constant volume (cv).
Less than
Equal to
More than
None of these
When a system changes its state from one equilibrium state to another equilibrium state, then the path of successive states through which the system has passed, is known as
Thermodynamic law
Thermodynamic process
Thermodynamic cycle
None of these
The isothermal and adiabatic processes are regarded as
Reversible process
Irreversible process
Reversible or irreversible process
None of these
The atomic mass of oxygen is
12
14
16
32
When a body is subjected to a direct tensile stress (σx) in one plane accompanied by a simple shear stress (τxy), the minimum normal stress is
(σx/2) + (1/2) × √(σx² + 4 τ²xy)
(σx/2) - (1/2) × √(σx² + 4 τ²xy)
(σx/2) + (1/2) × √(σx² - 4 τ²xy)
(1/2) × √(σx² + 4 τ²xy)
The mass of carbon per kg of flue gas is given by
(11/3) CO2 + (3/7) CO
(3/7) CO2 + (11/3) CO
(7/3) CO2 + (3/11) CO
(3/11) CO2 + (7/3) CO
The total elongation produced in a bar of uniform section hanging vertically downwards due to its own weight is equal to that produced by a weight
Of same magnitude as that of bar and applied at the lower end
Half the weight of bar applied at lower end
Half of the square of weight of bar applied at lower end
One fourth of weight of bar applied at lower end
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
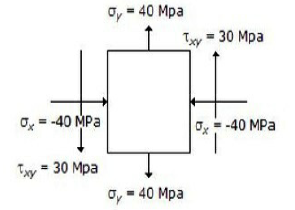
10 MPa
30 MPa
50 MPa
100 MPa
The heat energy stored in the gas and used for raising the temperature of the gas is known as
External energy
Internal energy
Kinetic energy
Molecular energy
