For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
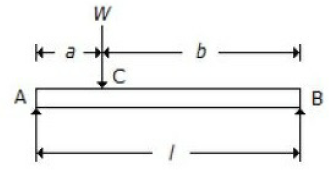
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
Correct Answer :
B. Wa²b²/3EIl
Related Questions
The total strain energy stored in a body is termed a
Resilience
Proof resilience
Modulus of resilience
Toughness
Reversed Joule cycle is known as
Carnot cycle
Bell-Coleman cycle
Rankine cycle
Stirling cycle
The cycle in which heat is supplied at constant volume and rejected at constant pressure is known as
Dual combustion cycle
Diesel cycle
Atkinson cycle
Rankine cycle
Otto cycle is also known as
Constant pressure cycle
Constant volume cycle
Constant temperature cycle
Constant temperature and pressure cycle
The processes occuring in open system which permit the transfer of mass to and from the system, are known as
Flow processes
Non-flow processes
Adiabatic processes
None of these
In a prismatic member made of two materials so joined that they deform equally under axial stress, the unit stresses in two materials are
Equal
Proportional to their respective moduli of elasticity
Inversely proportional to their moduli of elasticity
Average of the sum of moduli of elasticity
The property of a material which allows it to be drawn into a smaller section is called
Plasticity
Ductility
Elasticity
Malleability
The absolute zero pressure can be attained at a temperature of
0°C
273°C
273 K
None of these
Which of the following gas is mostly used in town for street and domestic lighting and heating?
Producer gas
Coal gas
Mond gas
Coke oven gas
The most probable velocity of the gas molecules is given by
√(KT/m)
√(2KT/m)
√(3KT/m)
√(5KT/m)
A riveted joint in which every rivet of a row is opposite to other rivet of the outer row, is known as
Chain riveted joint
Diamond riveted joint
Crisscross riveted joint
Zigzag riveted joint
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
The deformation per unit length is called
Tensile stress
Compressive stress
Shear stress
Strain
The shear force diagram for a simply supported beam carrying a uniformly distributed load of w per unit length, consists of
One right angled triangle
Two right angled triangles
One equilateral triangle
Two equilateral triangles
When a rectangular beam is loaded transversely, the maximum compressive stress is developed on the
Top layer
Bottom layer
Neutral axis
Every cross-section
In closed cycle gas turbine, the air is compressed
Isothermally
Isentropically
Polytropically
None of these
When it is indicated that a member is elastic, it means that when force is applied, it will
Not deform
Be safest
Stretch
Not stretch
When a body is subjected to two equal and opposite pushes, as a result of which the body tends to reduce its length, then
The stress and strain induced is compressive
The stress and strain induced is tensile
Both A and B is correct
None of these
If a material expands freely due to heating it will develop
Thermal stresses
Tensile stress
Bending
No stress
According to Kelvin-Planck's statement, a perpetual motion of the __________ is impossible.
First kind
Second kind
Third kind
None of these
The natural petroleum may be separated into
Petrol
Kerosene
Fuel oil
Lubricating oil
A close cycle gas turbine gives __________ efficiency as compared to an open cycle gas turbine.
Same
Lower
Higher
None of these
The mass of excess air supplied is equal to
(23/100) × Mass of excess carbon
(23/100) × Mass of excess oxygen
(100/23) × Mass of excess carbon
(100/23) × Mass of excess oxygen
Energy can neither be created nor destroyed, but it can be transformed from one form to another. This statement is known as
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kinetic theory of gases
Charles' law states that all perfect gases change in volume by __________ of its original volume at 0°C for every 1°C change in temperature, when pressure remains constant.
1/27th
1/93th
1/173th
1/273th
The main cause for the irreversibility is
Mechanical and fluid friction
Unrestricted expansion
Heat transfer with a finite temperature difference
All of the above
The specific heat at constant volume is
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant pressure
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
The amount of heat required to raise the temperature of 1 kg of water through one degree
Any one of the above
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
The efficiency of Joule cycle is
Greater than Carnot cycle
Less than Carnot cycle
Equal to Carnot cycle
None of these
The smallest quantity of a substance, which can exist by itself in a chemically recognizable form is known as
Element
Compound
Atom
Molecule
