Related Questions
The charge on an atom of is
+40
+27
+14
Zero
The interrelationship between matter and energy was given by
Rutherford
Joule
de Broglie
Einstein
Which of the following has the highest mass?
Neutron
Alpha particle
Electron
Deuterium
In an atom of hydrogen, which of the following orbitais has the lowest energy for an electron present in it?
3r
2p
4p
2s
The number of unpaired electrons in the ground state of chromium is
1
6
7
2
The angular momentum of the moving electron is equal to
The relationship between energy of a radiation and its frequency was given by
Planck
Rutherford
Einstein
Joule
For a given energy level the number of orbitals is equal to
Bohr's concept of the orbit in an atom was contradicted by
de Broglie relationship
Uncertainty principle
Planck's hypothesis
Hund's rule
rays are a stream of
He
electrons
positron
neutrons
Which of the following weighs the least?
1 g of
2 litres of at N.T.P.
molecules of
atoms of carbon
The fundamental particle that has least mass is
meson
alpha-particle
electron
neutron
The atomic number of an element having the valence shell electronic configuration is
35
26
23
34
Which elements is represented by the following electronic configuration?
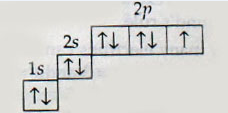
Nitrogen
Oxygen
Fluorine
Neon
The maximum number of electrons that can be accommodated by an atom in g-sub-energy level are
20
25
18
12
The number of unpaired electrons in fluorine atoms is/are
4
2
1
0
Cathode rays contain a stream of
The wavelength of an electron
is equal to that of light
remains constant with velocity
decreases with an increasing velocity
increases with an decreasing velocity
Argon is isoelectronic with
In the case of atomic spectrum of hydrogen which series of lines lie in the visible region?
Balmer
Paschen
Pfund
None of these
The ion that is isoelectronic with CO is
Among the following ions, which are has the highest paramagnetism?
The number of neutrons present in are
28
39
20
19
Isotones have same
mass number
nuclear mass
no. of neutrons
no. of electrons
A dipositive ion has in the K shell, 8 e~ s in the L shell and in the M shell. Atomic number of Z is
19
20
16
15
The number of neutrons in the deuterium atom is
16
3
1
0
The atomic structure of an atom of an element is Is . The element is
Cl-35
S-32
N-18
O-16
Which of the following atoms has a non-spherical outermost orbital.
H
Li
Be
B
When the electron is excited from K level to M level we get
- rays
cathode rays
continuous spectra
absorption spectra
No two electrons in an atom can have the same values of all four quantum numbers according to
Hund's rules
Flemming rule
Pauli's exclusion principle
Bohr theory
