For the irreversible reaction, Ca+ 2C = Ca C2 , Δ H°298 = - 60000 J . mole-1 . If a system initially containing 2 moles of calcium, 3 moles of carbon and 1 mole of calcium carbide is allowed to react to completion, the heat evolved at 298 K will be
30,000 J
60,000 J
90,000 J
2,40,000 J
Correct Answer :
C. 90,000 J
Related Questions
A steam carrying pipeline is insulated with two layers of insulating materials with the inferior insulation material forming the inner part. If the two insulating layers are interchanged, the heat conducted will
Increase
Decrease
Remain unaffected
May increase or decrease; depends on the thickness of each layer
The ratio of mass of a neutron to that of an electron is about 1839. What is the ratio of the mass of a proton to that of an electron?
159
1837
2537
10000
Viscoelastic behaviour is observed in __________ materials.
Non-crystalline organic polymeric
Ceramic
All crystalline
All amorphous
A jet engine turbine blade is normally manufactured by
Forging
Shell moulding
Investment casting
Pressure die casting
Consideration of the creep is the most important in case of the
Blades of gas turbine
Piston of an I. C. engine
Flywheel of steam engine
Cycle chain
The 'laughing gas' is
Nitrous oxide
Nitric oxide
Nitrogen trioxide
Nitrogen pentoxide
Which of the following contributes maximum as main source of sulphur in the blast furnace charge?
Sinter
Coke
Iron ore
Limestone
Faraday's law of electrolysis is related to the
Cation speed
Atomic number of the cation
Equivalent mass of the electrolyte
None of these
Basicity 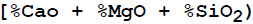 of the slag in Indian blast furnace is in the range of
of the slag in Indian blast furnace is in the range of
0.7-1.0
1.1-1.4
1.5 - 1.8
2.0 - 2.5
In the X-ray radiography technique, the tube voltage for thicker plates as compared to thin plates, should be
Higher, as it gives higher wavelength
Lower, as it gives higher wavelength
Higher, as it gives shorter wavelength
Lower, as it gives shorter wavelength
Suitability of steel for its use in cable is judged by its strength in
Tension
Compression
Torsion
Shear
The trace metal present in insulin is
Cu
Zn
Fe
Ni
Thermodynamic cycle involved in the working of a thermal power plant is the __________ cycle.
Joule
Carnot
Rankine
Brayton
In a good rimming steel
Carbon and silicon should be low
Silicon should be low but carbon should be high
Both silicon & carbon should be high
Silicon should be high but carbon should be low
Minimum number of members required to form a Public Limited Joint Stock Com-pany is
7
10
12
17
Chromium molybdenum steel cannot be welded using __________ welding.
Thermit
Electrical resistance
Oxy-acetylene
Any of these
If a solid is compressed adiabatically in its elastic range, its __________ remains constant
Internal energy
Enthalpy
Entropy
Temperature
Which of the following relationships is correct for relating the three elastic constants of an isotropic elastic material (where, E = Young's modulus, G = Modulus of rigidity or shear modulus v = Poisson's ratio)?
E = 2G (1 + v)
E = G (1 + v)
E = G (1 + v)/2
E = 2G (1 + 2v)
Material of construction of the electrode used in the electric resistance welding is
Stainless steel
Graphite
Copper
Steel
Blow off cock is provided in steam boiler to
Guard the boiler in case of abnormal rise in steam pressure
Drain out water and sludge during its repair
Regulate the supply of water in boiler
Put out the fire in the furnace when the water level in the boiler falls to unsafe limit
Joule-Thomson co-efficient is the ratio of
Pressure change to temperature change occuring during adiabatic compression of a gas
Pressure change to temperature change occuring during adiabatic throttling of a gas
Temperature change to pressure change occuring during adiabatic compression of a gas
Temperature change to pressure change occuring during adiabatic throttling of a gas
Rankine cycle comprises of two isothermal and two __________ processes.
Isobaric
Polytropic
Isentropic
None of these
Dephosphorization of molten pig iron is favoured by
Oxidising and basic slag
Reducing and basic slag
High activity co-efficient of phosphorous in metal
Oxidising and neutral slag
Which of the following metals is the most prone to work hardening?
Brass
Aluminium
Copper
Lead
Drills are usually made of
Cermets
High speed steel
Alloy steel
Tungsten carbide
Starting friction is low in case of the __________ lubrication.
Boundary
Hydrodynamic
Hydrostatic
Mixed/semi-fluid
Air/fuel ratio by weight for combustion of methane with theoretical quantity of air will be about
9 : 1
17 : 1
23 : 1
29 : 1
Gas turbine normally employs a constant __________ cycle.
Pressure
Temperature
Volume
None of these
A gas which is collected over water becomes moistened due to water vapor, exerts its own partial pressure at the gas temperature, which is termed as its
Aqueous tension
Saturated humidity
Vapor pressure
Absolute humidity
Plants produce carbohydrates from the CO2 present in the atmosphere by
Polymerisation
Photochemical reaction
Oxidation
None of these
