For the reversible exothermic reaction, N2 + 3H2 2NH3, increase of pressure would
Shift the equilibrium towards right
Give higher yield of NH3
Both (B) and (C)
Neither (A) nor (B)
Correct Answer :
C. Both (B) and (C)
Related Questions
A system in which there is exchange of energy but not of mass, is called a/an __________ system.
Isolated
Open
Insulated
Closed
At the absolute zero temperature, the entropy of every perfectly crystalline substance becomes zero. This follows from the
Third law of thermodynamics
Second law of thermodynamics
Nernst heat theorem
Maxwell's relations
The necessary condition for phase equilibrium in a multiphase system of N components is that the
Chemical potentials of a given component should be equal in all phases
Chemical potentials of all components should be same in a particular phase
Sum of the chemical potentials of any given component in all the phases should be the same
None of these
Entropy change of the reaction, H2O (liquid) → H2O (gas), is termed as the enthalpy of
Solution
Vaporisation
Formation
Sublimation
Which of the following is not an extensive property?
Free energy
Entropy
Refractive index
None of these
Which of the following is not an intensive property?
Chemical potential
Surface tension
Heat capacity
None of these
It is desired to bring about a certain change in the state of a system by performing work on the system under adiabatic conditions.
The amount of work needed is path dependent
Work alone cannot bring out such a change of state
The amount of work needed is independent of path
More information is needed to conclude anything about the path dependence or otherwise of the work needed
Equilibrium constant decreases as the temperature
Increases, for an exothermic reaction
Decreases, for an exothermic reaction
Increases, for an endothermic reaction
None of these
__________ decreases during adiabatic throttling of a perfect gas.
Entropy
Temperature
Enthalpy
Pressure
Maximum work that could be secured by expanding the gas over a given pressure range is the __________ work.
Isothermal
Adiabatic
Isentropic
None of these
The Carnot co-efficient of performance (COP) of a domestic air conditioner compared to a household refrigerator is
Less
More
Same
Dependent on climatic conditions
For an ideal gas, the activity co-efficient is
Directly proportional to pressure
Inversely proportional to pressure
Unity at all pressures
None of these
Which of the following identities can be most easily used to verify steam table data for superheated steam?
(∂T/∂V)S = (∂p/∂S)V
(∂T/∂P)S = (∂V/∂S)P
(∂P/∂T)V = (∂S/∂V)T
(∂V/∂T)P = -(∂S/∂P)T
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
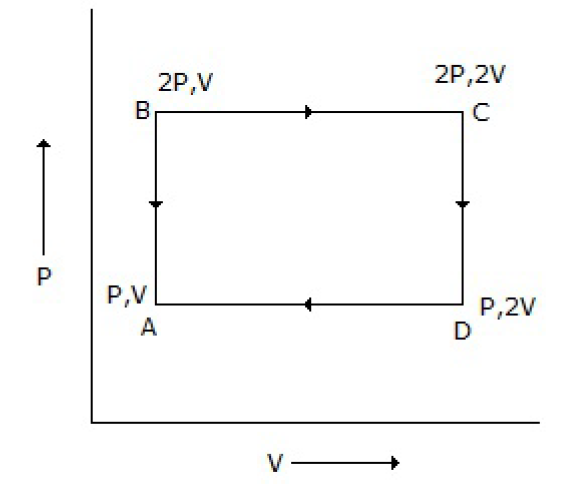
PV
2PV
PV/2
0
In Joule-Thomson porous plug experiment, the
Enthalpy does not remain constant
Entire apparatus is exposed to surroundings
Temperature remains constant
None of these
Number of components (C), phase (P) and degrees of freedom (F) are related by Gibbs phase rule as
P + F - C = 2
C = P - F + 2
F = C - P - 2
P = F - C - 2
A thermodynamic system is taken from state A to B along ACB and is brought back to A along BDA as shown below in the P-V diagram. The net work done during the complete cycle is given by the area covered by

P1ACBP2P1
ACBB1A1A
ACBDA
ADBB1A1A
The expression for entropy change, ΔS = n Cp . ln (T2/T1), is valid for the __________ of a substance.
Simultaneous pressure & temperature change
Heating
Cooling
Both (B) and (C)
Dryness fraction of wet steam is defined as the ratio of mass of vapour in the mixture to the mass of mixture __________ calorimeter is not used for measuring the dryness fraction of steam.
Bomb
Separating
Bucket
Throttling
__________ increases with increase in pressure.
The melting point of wax
The boiling point of a liquid
Both (A) and (B)
Neither (A) nor (B)
At 60° C, vapour pressure of methanol and water are 84.562 kPa and 19.953 kPa respectively. An aqueous solution of methanol at 60° C exerts a pressure of 39.223 kPa; the liquid phase and vapour phase mole fractions of methanol are 0.1686 and 0.5714 respectively. Activity co-efficient of methanol is
1.572
1.9398
3.389
4.238
The molar excess Gibbs free energy, gE, for a binary liquid mixture at T and P is given by, (gE/RT) = A . x1. x2, where A is a constant. The corresponding equation for ln y1, where y1 is the activity co-efficient of component 1, is
A . x22
Ax1
Ax2
Ax12
The melting point of paraffin wax (which contracts on solidification) __________ with pressure rise.
Increases
Decreases
Remains unchanged
Decreases linearly
Henry's law is closely obeyed by a gas, when its __________ is extremely high.
Pressure
Solubility
Temperature
None of these
In an irreversible process
Tds = dE - dW = 0
dE - dW - Tds = 0
Tds - dE + dW < 0
Tds - dT + dW < 0
What is the degree of freedom for a system comprising liquid water equilibrium with its vapour?
0
1
2
3
The shape of T-S diagram for Carnot Cycle is a
Rectangle
Rhombus
Trapezoid
Circle
Gibbs phase rule finds application, when heat transfer occurs by
Conduction
Convection
Radiation
Condensation
In an ideal refrigeration cycle, the change in internal energy of the fluid is
+ve
-ve
0
Either of the above three; depends on the nature of refrigerant
Minimum number of phases that exists in a system is 1. Number of chemical species in a colloidal system is
1
2
3
4
