Gases are cooled in Joule-Thomson expansion, when it is __________ inversion temperature.
Below
At
Above
Either 'b' or 'c'
Correct Answer :
A. Below
Related Questions
Pick out the correct statement.
Like internal energy and enthalpy, the absolute value of standard entropy for elementary substances is zero
Melting of ice involves increase in enthalpy and a decrease in randomness
The internal energy of an ideal gas depends only on its pressure
Maximum work is done under reversible conditions
Linde gas liquefaction process employs cooling
By throttling
By expansion in an engine
At constant pressure
None of these
Consider the process A & B shown in the figure given below: In this case, it is possible that
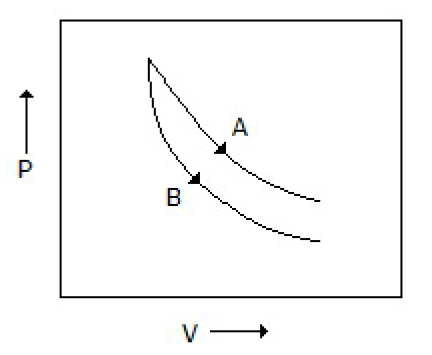
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
In which of the following reaction equilibrium, the value of equilibrium constant Kp will be more than is Kc?
2HI H2 + I2
N2O4 2NO2
2SO2 + O2 2SO3
None of these
Specific volume of an ideal gas is
Equal to its density
The reciprocal of its density
Proportional to pressure
None of these
Dryness fraction of wet steam is defined as the ratio of mass of vapour in the mixture to the mass of mixture __________ calorimeter is not used for measuring the dryness fraction of steam.
Bomb
Separating
Bucket
Throttling
The root mean square speed of molecules of a gas is equal to (where, m = mass of the molecule K = Boltzmanns constant, T = absolute temperature)
√(2KT/m)
√(3KT/m)
√(6KT/m)
3KT/m
Gibbs free energy (F) is defined as
F = E - TS
F = H - TS
F = H + TS
F = E + TS
The variation of heat of reaction with temperature at constant pressure is given by the __________ law.
Kelvin's
Antoines
Kirchoffs
None of these
Compressibility factor (i.e., the ratio of actual volume of gas to the volume predicted by ideal gas law) for all gases are
Always greater than one
Same at the same reduced temperature
Same at the same reduced pressure
Both (B) & (C)
In an ideal gas mixture, fugacity of a species is equal to its
Vapor pressure
Partial pressure
Chemical potential
None of these
The equilibrium constant for a chemical reaction at two different temperatures is given by
Kp2/Kp1 = - (ΔH/R) (1/T2 - 1/T1)
Kp2/Kp1 = (ΔH/R) (1/T2 - 1/T1)
Kp2/Kp1 = ΔH (1/T2 - 1/T1)
Kp2/Kp1 = - (1/R) (1/T2 - 1/T1)
The difference between isothermal compressibility and adiabatic compressibility for an ideal gas is
0
+ve
-ve
∞
Number of components (C), phase (P) and degrees of freedom (F) are related by Gibbs phase rule as
P + F - C = 2
C = P - F + 2
F = C - P - 2
P = F - C - 2
Ideal refrigeration cycle is
Same as Carnot cycle
Same as reverse Carnot cycle
Dependent on the refrigerant's properties
The least efficient of all refrigeration processes
Specific heat of a gas for a reversible adiabatic process is
Negative
Zero
Infinity
None of these
The expression for entropy change, ΔS = n Cp . ln (T2/T1), is valid for the __________ of a substance.
Simultaneous pressure & temperature change
Heating
Cooling
Both (B) and (C)
Which of the following is not an extensive property?
Free energy
Entropy
Refractive index
None of these
What happens in a reversible adiabatic compression?
Heating occurs
Cooling occurs
Pressure is constant
Temperature is constant
What is the value of ln y (where y = activity co-efficient) for ideal gases?
Zero
Unity
Infinity
Negative
Equilibrium constant decreases as the temperature
Increases, for an exothermic reaction
Decreases, for an exothermic reaction
Increases, for an endothermic reaction
None of these
Tea kept in a thermos flask is vigorously shaken. If the tea is considered as a system, then its temperature will
Increase
Decrease
Remain unchanged
First fall and then rise
Law of corresponding states says that
Two different gases behave similarly, if their reduced properties (i.e. P, V and T) are same
The surface of separation (i. e. the meniscus) between liquid and vapour phase disappears at the critical temperature
No gas can be liquefied above the critical temperature, howsoever high the pressure may be.
The molar heat of energy of gas at constant volume should be nearly constant (about 3 calories)
In the reaction, H2 +I2 2HI, addition of an inert gas will
Increase the partial pressure of H2
Increase the partial pressure of I2
Increase the total pressure and hence shift the equilibrium towards the right
Not affect the equilibrium conditions
Variation of equilibrium pressure with temperature for any two phases of a given substances is given by the __________ equation.
Gibbs-Duhem
Maxwell's
Clapeyron
None of these
A refrigerator works on the principle of __________ law of thermodynamics.
Zeroth
First
Second
Third
The expression, nCv(T2 - T1), is for the __________ of an ideal gas.
Work done under adiabatic condition
Co-efficient of thermal expansion
Compressibility
None of these
__________ explains the equilibrium constant for any chemical reaction.
Henry's law
Law of mass action
Hess's law
None of these
Degree of freedom of a system consisting of a gaseous mixture of H2 and NH3 will be
0
1
2
3
When dilute aqueous solutions of two salts are mixed, the process is associated with
Decrease in temperature
Increase in temperature
No change in temperature
Change in temperature which is a function of composition
