Joule-Thomson co-efficient which is defined as, η = (∂T/∂P)H = 1/Cp (∂H/∂T)P, changes sign at a temperature known as inversion temperature. The value of Joule-Thomson co-efficient at inversion temperature is
0
∞
+ve
-ve
Correct Answer :
A. 0
Related Questions
Critical temperature is defined as the temperature above which a gas will
Not liquify (barring exceptions)
Immediately liquify
Never liquify however high the pressure may be
None of these
If different processes are used to bring about the same chemical reaction, the enthalpy change is same for all of them. This is __________ law.
Hess's
Kirchoff's
Lavoisier and Laplace
None of these
Critical solution temperature (or the consolute temperature) for partially miscible liquids (e.g., phenol-water) is the minimum temperature at which
A homogeneous solution (say of phenol water) is formed
Mutual solubility of the two liquids shows a decreasing trend
Two liquids are completely separated into two layers
None of these
In any spontaneous process,
Only F decreases
Only A decreases
Both F and A decreases
Both F and A increase
Compressibility factor (i.e., the ratio of actual volume of gas to the volume predicted by ideal gas law) for all gases are
Always greater than one
Same at the same reduced temperature
Same at the same reduced pressure
Both (B) & (C)
Third law of thermodynamics is helpful in
Prediction of the extent of a chemical reaction
Calculating absolute entropies of substances at different temperature
Evaluating entropy changes of chemical reaction
Both (B) and (C)
All gases except __________ shows a cooling effect during throttling process at atmospheric temperature and pressure.
Oxygen
Nitrogen
Air
Hydrogen
The co-efficient of performance (COP) of a refrigerating system, which is its index of performance, is defined as the ratio of useful refrigeration to the net work. The units of __________ and COP are the same.
Kinematic viscosity
Work
Temperature
None of these
The point at which all the three (solid, liquid and gas) phases co-exist, is known as the __________ point.
Freezing
Triple
Boiling
Boyle
In the equation, PVn = constant, if the value of n is in between 1 and y (i.e. Cp/Cv), then it represents a reversible __________ process.
Isometric
Polytropic
Isentropic
Isobaric
In case of a reversible process (following pvn = constant), work obtained for trebling the volume (v1 = 1 m3 and v23 m3) is maximum, when the value of 'n' is
0
1
y = 1.44
1.66
At a given temperature, the volume of a gas dissolved in a solvent __________ with increase in pressure.
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the gas
Joule-Thomson co-efficient for a perfect gas is
Zero
Positive
Negative
None of these
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
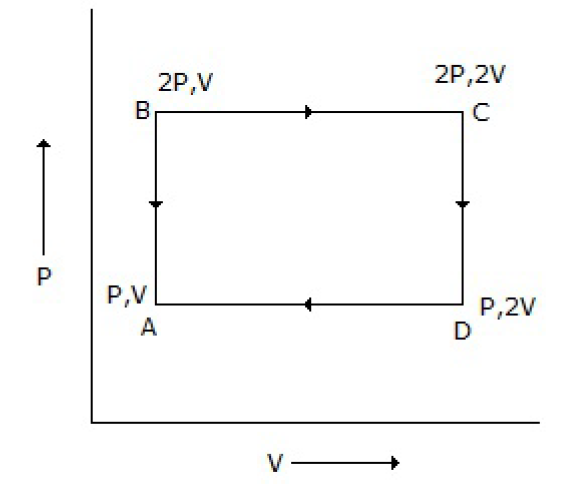
PV
2PV
PV/2
0
A large iceberg melts at the base, but not at the top, because of the reason that
Ice at the base contains impurities which lowers its melting point
Due to the high pressure at the base, its melting point reduces
The iceberg remains in a warmer condition at the base
All (A), (B) and (C)
The most important application of distribution law is in
Evaporation
Liquid extraction
Drying
Distillation
Pick out the wrong statement.
Trouton's ratio of non-polar liquids is calculated using Kistyakowsky equation
Thermal efficiency of a Carnot engine is always less than 1
An equation relating pressure, volume and temperature of a gas is called ideal gas equation
None of these
The internal energy of a gas obeying P (V - b) RT (where, b is a positive constant and has a constant Cv), depends upon its
Pressure
Volume
Temperature
All (A), (B) & (C)
In reactions involving solids and liquids (where change in volume is negligible), the heat of reaction at constant pressure as compared to that at constant volume is
More
Less
Same
Unpredictable; depends on the particular reaction
At equilibrium condition, the chemical potential of a material in different phases in contact with each other is equal. The chemical potential for a real gas (μ) is given by (where, μ = standard chemical potential at unit fugacity (f° = 1 atm.) and the gas behaves ideally.)
μ° + RT ln f
μ°+ R ln f
μ° + T ln f
μ° + R/T ln f
Melting of wax is accompanied with __________ in entropy.
Increase
Decrease
No change
None of these
A Carnot cycle consists of the following steps:
Two isothermal and two isentropic
Two isobaric and two isothermal
Two isochoric and two isobaric
Two isothermals and two isochoric
Keeping the pressure constant, to double the volume of a given mass of an ideal gas at 27°C, the temperature should be raised to __________ °C.
270
327
300
540
(∂E/∂T)V is the mathematical expression for
CV
Enthalpy change
Free energy change
None of these
In polytropic process (PVn = constant), if n = 1; it means a/an __________ process.
Adiabatic
Reversible
Isothermal
None of these
For a constant volume process __________ by the system is used only to increase the internal energy.
Heat absorbed
Work done
Both (A) & (B)
Neither (A) nor (B)
Heat is added at constant pressure in an ideal __________ cycle.
Stirling
Brayton
Rankine
Both (B) and (C)
In a working refrigerator, the value of COP is always
0
< 0
< 1
> 1
Boiling of liquid is accompanied with increase in the
Vapor pressure
Specific Gibbs free energy
Specific entropy
All (A), (B) and (C)
__________ decreases during adiabatic throttling of a perfect gas.
Entropy
Temperature
Enthalpy
Pressure
