Modulus of rigidity is defined as the ratio of
Longitudinal stress to longitudinal strain
Volumetric stress to volumetric strain
Lateral stress to Lateral strain
Shear stress to shear strain
Correct Answer :
D. Shear stress to shear strain
Related Questions
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
Otto cycle consists of
Two constant volume and two isentropic processes
Two constant pressure and two isentropic processes
Two constant volume and two isothermal processes
One constant pressure, one constant volume and two isentropic processes
A column of length (l) with both ends fixed may be considered as equivalent to a column of length __________ with both ends hinged.
l/8
l/4
l/2
l
If a material expands freely due to heating it will develop
Thermal stresses
Tensile stress
Bending
No stress
An isothermal process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The thermodynamic difference between a Rankine cycle working with saturated steam and the Carnot cycle is that
Carnot cycle can't work with saturated steam
Heat is supplied to water at temperature below the maximum temperature of the cycle
A Rankine cycle receives heat at two places
Rankine cycle is hypothetical
The value of cp/cv for air is
1
1.4
1.45
2.3
The extremeties of any diameter on Mohr's circle represent
Principal stresses
Normal stresses on planes at 45°
Shear stresses on planes at 45°
Normal and shear stresses on a plane
A composite shaft consisting of two stepped portions having spring constants K₁ and K₂ is held between two rigid supports at the ends. Its equivalent spring constant is
K₁ K₂
(K₁ + K₂)/ 2
(K₁ + K₂)/ K₁ K₂
K₁ K₂/ (K₁ + K₂)
The strain energy stored in a body due to suddenly applied load compared to when it is applied gradually is
Same
Twice
Four times
Eight times
Reversed joule cycle is called
Carnot cycle
Rankine cycle
Brayton cycle
Bell Coleman cycle
The stress necessary to initiate yielding is
Considerably greater than that necessary to continue it
Considerably lesser than that necessary to continue it
Greater than that necessary to stop it
Lesser than that necessary to stop it
The ideal efficiency of a Brayton cycle with regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
A column that fails due to direct stress, is called
Short column
Long column
Weak column
Medium column
A cycle consisting of two constant pressure and two isentropic processes is known as
Carnot cycle
Stirling cycle
Otto cycle
None of these
High air-fuel ratio in gas turbines
Increases power output
Improves thermal efficiency
Reduces exhaust temperature
Do not damage turbine blades
Which of the following is the extensive property of a thermodynamic system?
Pressure
Volume
Temperature
Density
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then maximum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
The absolute zero temperature is taken as
237°C
-273°C
-237°C
273°C
The maximum bending moment for the beam shown in the below figure, is
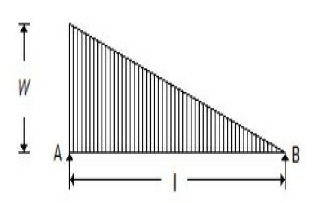
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
The shear force at the centre of a simply supported beam with a gradually varying load from zero at both ends to w per metre at the centre, is
Zero
wl/4
wl/2
wl²/2
The amount of heat generated per kg of fuel is known as
Calorific value
Heat energy
Lower calorific value
Higher calorific value
If Th is the torque resisting capacity of a hollow shaft and Ts is that of a solid shaft, of the same material, length and weight. Then,
Th > Ts
Th < Ts
Th = Ts
None of these
Energy can neither be created nor destroyed, but it can be transformed from one form to another. This statement is known as
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kinetic theory of gases
If the value of n = 0 in the equation pvn = C, then the process is called
Constant volume process
Adiabatic process
Constant pressure process
Isothermal process
If a part is constrained to move and heated, it will develop
Principal stress
Tensile stress
Compressive stress
Shear stress
Second law of thermodynamics defines
Heat
Work
Internal energy
Entropy
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
Tensile strength of a material is obtained by dividing the maximum load during the test by the
Area at the time of fracture
Original cross-sectional area
Average of (A) and (B)
Minimum area after fracture
