Molar heat capacity of water in equilibrium with ice at constant pressure is __________ Kcal/kg mole. °K
0
∞
50
100
Correct Answer :
B. ∞
Related Questions
Pick out the wrong statement.
Trouton's ratio of non-polar liquids is calculated using Kistyakowsky equation
Thermal efficiency of a Carnot engine is always less than 1
An equation relating pressure, volume and temperature of a gas is called ideal gas equation
None of these
In an ideal refrigeration cycle, the change in internal energy of the fluid is
+ve
-ve
0
Either of the above three; depends on the nature of refrigerant
For a multi-component system, the term chemical potential is equivalent to the
Molal concentration difference
Molar free energy
Partial molar free energy
Molar free energy change
Absorption/evolution of heat during conversion of a substance from one allotropic form to another is termed as the heat of
Sublimation
Fusion
Transition
Vaporisation
The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
What is the value of Joule-Thomson co-efficient for an ideal gas?
+ve
-ve
0
∞
Specific/molar Gibbs free energy for a pure substance does not change during
Sublimation
Vaporisation
Melting
Either (A), (B) or (C)
The equation, Cp - Cv = R, is true for __________ gas.
No
Any real
Only ideal
Both (B) and (C)
For any system, what is the minimum number of degrees of freedom?
0
1
2
3
The reaction A (l) → R(g) is allowed to reach equilibrium conditions in an autoclave. At equilibrium, there are two phases, one a pure liquid phase of A and the other a vapor phase of A, R and S. Initially A alone is present. The numbers of degrees of freedom are:
1
2
3
0
Pick out the Clausius-Clapeyron equation from the following:
dP/dT = ΔH/TΔV
ln P = - (ΔH/RT) + constant
ΔF = ΔH + T [∂(ΔF)/∂T]P
None of these
Henry's law is closely obeyed by a gas, when its __________ is extremely high.
Pressure
Solubility
Temperature
None of these
Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
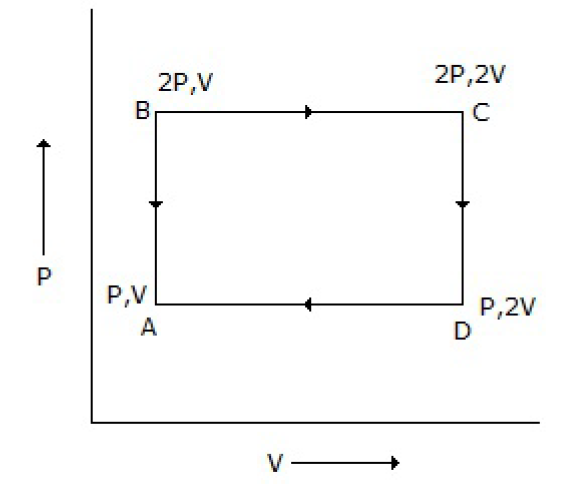
PV
2PV
PV/2
0
Which of the following is an undesirable characteristic of a refrigerant?
It should be non-explosive
It should have a sub-atmospheric vapor pressure at the temperature in refrigerator coils
Its vapor pressure at the condenser temperature should be very high
None of these
At equilibrium condition, the chemical potential of a material in different phases in contact with each other is equal. The chemical potential for a real gas (μ) is given by (where, μ = standard chemical potential at unit fugacity (f° = 1 atm.) and the gas behaves ideally.)
μ° + RT ln f
μ°+ R ln f
μ° + T ln f
μ° + R/T ln f
A refrigerator works on the principle of __________ law of thermodynamics.
Zeroth
First
Second
Third
Claude gas liquefaction process employs cooling
At constant pressure
By throttling
By expansion in an engine
None of these
What is the degree of freedom for a system comprising liquid water equilibrium with its vapour?
0
1
2
3
Minimum number of phases that exists in a system is 1. Number of chemical species in a colloidal system is
1
2
3
4
(1/V) (∂V/∂T)P is the mathematical expression
Joule-Thomson co-efficient
Specific heat at constant pressure (Cp)
co-efficient of thermal expansion
Specific heat at constant volume (CV)
Which of the following is a thermodynamic property of a system?
Concentration
Mass
Temperature
Entropy
Helmholtz free energy (A) is defined as
A = H - TS
A = E - TS
A = H + TS
None of these
Which of the following is not a reversible process?
Expansion of an ideal gas against constant pressure
Atmospheric pressure vaporisation of water at 100°C
Solution of NaCl in water at 50°C
None of these
For multi-component multiple phases to be in equilibrium at the same pressure and temperature, the __________ of each component must be same in all phases.
Chemical potential
Fugacity
Both (A) and (B)
Neither (A) nor (B)
The internal energy of an incompressible fluid depends upon its
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
The number of degrees of freedom at the triple point of water is
0
1
2
3
A closed system is cooled reversibly from 100°C to 50°C. If no work is done on the system
its internal energy (U) decreases and its entropy (S) increases
U and S both decreases
U decreases but S is constant
U is constant but S decreases
At the critical point of a substance
The surface tension vanishes
Liquid and vapour have the same density
There is no distinction between liquid and vapour phases
All (A), (B) and (C)
Free energy change at equilibrium is
Zero
Positive
Negative
Indeterminate
