Pick out the wrong statement:
The expansion of a gas in vacuum is an irreversible process
An isometric process is a constant pressure process
Entropy change for a reversible adiabatic process is zero
Free energy change for a spontaneous process is negative
Correct Answer :
B. An isometric process is a constant pressure process
Related Questions
Reduced pressure of a gas is the ratio of its
Pressure to critical pressure
Critical pressure to pressure
Pressure to pseudocritical pressure
Pseudocritical pressure to pressure
Pick out the wrong statement
Phase rule variables are intensive properties
Heat and work are both state function
The work done by expansion of a gas in vacuum is zero
CP and CV are state function
One mole of nitrogen at 8 bar and 600 K is contained in a piston-cylinder arrangement. It is brought to 1 bar isothermally against a resisting pressure of 1 bar. The work done (in Joules) by the gas is
30554
10373
4988.4
4364.9
The total change in the enthalpy of a system is independent of the
Number of intermediate chemical reactions involved
Pressure and temperature
State of combination and aggregation in the beginning and at the end of the reaction
None of these
With increase in temperature, the internal energy of a substance
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the substance
Consider the process A & B shown in the figure given below: In this case, it is possible that
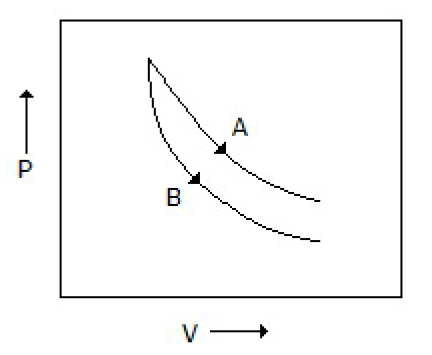
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
Near their critical temperatures, all gases occupy volumes __________ that of the ideal gas.
Less than
Same as
More than
Half
When dilute aqueous solutions of two salts are mixed, the process is associated with
Decrease in temperature
Increase in temperature
No change in temperature
Change in temperature which is a function of composition
Solubility of a substance which dissolves with an increase in volume and liberation of heat will be favoured by the
Low pressure and high temperature
Low pressure and low temperature
High pressure and low temperature
High pressure and high temperature
Pick out the wrong statement.
The values of (∂P/∂V)T and (∂2P/∂V2)T are zero for a real gas at its critical point
Heat transferred is equal to the change in the enthalpy of the system, for a constant pressure, non-flow, mechanically reversible process
Thermal efficiency of a Carnot engine depends upon the properties of the working fluid besides the source & sink temperatures
During a reversible adiabatic process, the entropy of a substance remains constant
An ideal gas is taken around the cycle ABCA as shown in P-V diagram below: The work done by the gas during the cycle is equal to

12 P1V1
6 P1 V1
3 P1V1
P1 V1
The equation, PV = nRT, is best obeyed by gases at
Low pressure & high temperature
High pressure & low temperature
Low pressure & low temperature
None of these
What happens in a reversible adiabatic expansion process?
Heating takes place
Cooling takes place
Pressure is constant
Temperature is constant
Joule-Thomson co-efficient for a perfect gas is
Zero
Positive
Negative
None of these
A reasonably general expression for vapourliquid phase equilibrium at low to moderate pressure is φi yi P = Yi xifi° where, Φ is a vapor fugacity component, Yi is the liquid activity co-efficient and fi° is the fugacity of the pure component i. the Ki value (Yi = Ki xi) is therefore, in general a function of
Temperature only
Temperature and pressure only
Temperature, pressure and liquid composition xi only
Temperature, pressure, liquid composition xi and vapour composition yi
As the temperature is lowered towards the absolute zero, the value of the quantity (∂ΔF/∂T) approaches
Zero
Unity
Infinity
None of these
Translational kinetic energy of molecules of an ideal gas is proportional to (where, T = absolute temperature of the gas)
T
√T
T2
1/√T
The theoretical minimum work required to separate one mole of a liquid mixture at 1 atm, containing 50 mole % each of n- heptane and noctane into pure compounds each at 1 atm is
-2 RT ln 0.5
-RT ln 0.5
0.5 RT
2 RT
In the reaction, H2 +I2 2HI, addition of an inert gas will
Increase the partial pressure of H2
Increase the partial pressure of I2
Increase the total pressure and hence shift the equilibrium towards the right
Not affect the equilibrium conditions
Mollier chart is a __________ plot.
Pressure vs. enthalpy
Pressure vs. volume
Enthalpy vs. entropy
Temperature vs. entropy
For a stable phase at constant pressure and temperature, the fugacity of each component in a binary system __________ as its mole fraction increases.
Decreases
Increases
Remain same
Decreases linearly
Pick out the correct equation relating 'F' and 'A'.
F = A + PV
F = E + A
F = A - TS
F = A + TS
If the vapour pressure at two temperatures of a solid phase in equilibrium with its liquid phase are known, then the latent heat of fusion can be calculated by the
Maxwell's equation
Clausius-Clapeyron Equation
Van Laar equation
Nernst Heat Theorem
A closed system is cooled reversibly from 100°C to 50°C. If no work is done on the system
its internal energy (U) decreases and its entropy (S) increases
U and S both decreases
U decreases but S is constant
U is constant but S decreases
Air enters an adiabatic compressor at 300K. The exit temperature for a compression ratio of 3, assuming air to be an ideal gas (Y = Cp/Cv = 7/5) and the process to be reversible, is
300 × (32/7)
300 × (33/5)
300 × (333/7)
300 × (35/7)
The heat capacities for the ideal gas state depend upon the
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
A solid is transformed into vapour without going to the liquid phase at
Triple point
Boiling point
Below triple point
Always
Pick out the correct statement.
Compression ratio of an Otto engine is comparatively higher than a diesel engine
Efficiency of an Otto engine is higher than that of a diesel engine for the same compression ratio
Otto engine efficiency decreases with the rise in compression ratio, due to decrease in work produced per quantity of heat
Diesel engine normally operates at lower compression ratio than an Otto engine for an equal output of work
When a system in equilibrium is subjected to a change in temperature, pressure or concentration, the equilibrium is displaced in a direction which tends to undo the effect of the change. This is called the
Le-Chatelier principle
Kopp's rule
Law of corresponding state
Arrhenius hypothesis
A system is said to be isopiestic, if there is no __________ change.
Temperature
Pressure
Volume
None of these
