Plasma is
A high temperature neutral gas
Nothing but ionised gas
A source of steady and highest controllable pressure
Formed at very low temperature
Correct Answer :
B. Nothing but ionised gas
Related Questions
Square steel key is normally strong in failure by shear & crushing. Keys are normally made of __________ steel bars.
Hot rolled mild
Cold rolled mild
Forged
Machinable stainless
Mho's scale of hardness, which consists of 10 standard minerals is used for the measurement of __________ hardness.
Scratch
Indentation
Dynamic
Rebound
Two solutions A1 & A2 have pH value of 2 & 6 respectively. It implies that the solution
A1 is more alkaline than solution A2
A1 is highly acidic
A2 is very slightly acidic
Both (B) & (C)
Wavelength of radiation emitted by a body depends on the __________ of its surface.
Temperature
Area
Nature
All 'a', 'b' & 'c'
__________ is the process of coating the surface of steel with aluminium oxide, thereby imparting it increased resistance to corrosion & oxidation upto a temperature of 700°C.
Veneering
Galvanising
Electroplating
Calorising
The ratio of mass of a neutron to that of an electron is about 1839. What is the ratio of the mass of a proton to that of an electron?
159
1837
2537
10000
Most important property of steels for use in automobile bodies is the
Formability
Yield strength
Toughness
Resilience
Which of the following varies as the square root of oil pressure during atomisation of fuel oil through a pressure jet burner?
Output & fineness
Velocity
Both (A) & (B)
None of these
__________ is used for tying the steel columns to concrete foundation.
Grouting
Eyebolt
Anchor bolts
Refractory castables
Pressure required to increase the density of water by about 1% is __________ atmosphere.
10
50
200
1000
Oxygen cylinders used for autogenous (cutting/welding) purposes are
Seamless & made of steel
Heat treated before use
Made by sand casting
Welded steel cylinders
The escape velocity of a body on earth which is independent of its mass is about __________ km/second.
3
7
11
15
Use of economiser in a boiler plant reduces the fuel consumption for steam generation by about __________ percent.
1
10
30
50
During decarburising of a plain carbon steel, the thickness of ferrite layer growth is proportional to
Time
Square root of time
Square of time
Cube of time
A high pressure boiler generates steam at a pressure greater than __________ kg/cm2.
10
30
50
80
For a small scale toy factory, the fixed cost per month is Rs. 5000/-. The variable cost per toy is Rs. 20 and sales price is Rs. 30 per toy. The break even production per month will be __________ toys.
250
500
1000
3000
The usual energy consumption in electric arc furnace steel making is __________ KWh/ton of steel.
60 - 100
400 - 700
1200 -1500
2000 - 2300
As the fluid flow rate increases, the float of the Rotameter
Rises in the tube
Rotates at higher speed
Rotates at lower speed
Drops down in the tube
Isotropic materials have the same __________ in all directions.
Induced stresses
Density
Elastic properties
Thermal properties
Nuclear fission of one atom of uranium-235 produces the energy equivalent to about __________ MeV.
20
200
500
2000
The thickness of oxide film is y at time t. If 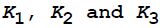 are the temperature dependent constants, the parabolic law of oxidation is given by
are the temperature dependent constants, the parabolic law of oxidation is given by
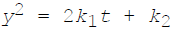
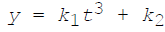
The two elements required to form substitutional solid solution should not have
Same crystalline structure
Same valency
Widely differing electronegativity
Same atomic sizes
Angular displacement can be measured suitably by a
Shaft encoder
Linear variable differential transducer (LVDT)
Stroboscope
Tachometer
Cobalt - 60 is used as a source of __________ in medical therapy & industrial radiography.
X-rays
γ-rays
α-rays
β-rays
Velocity of a gas in sound is not proportional to (where, T = Absolute temperature of the gas. P = Absolute pressure of the gas. y = Ratio of specific heats (Cp/Cv) ρ = specific weight of the gas)
√T
1/√P
√y
1/√ρ
For a first order chemical reaction, the concentration of the reactant decreases __________ with time.
Linearly
Exponentially
Logarithmically
Inversely
Calorific value of __________ are almost same.
Lignite & anthracite coal
Lignite & coal gas
Petrol & diesel
Coal gas & natural gas
Electrometallurgy is not involved in the extraction of __________ from its ore.
Aluminium
Tin
Lead
Both (B) & (C)
For the irreversible reaction, Ca+ 2C = Ca C2 , Δ H°298 = - 60000 J . mole-1 . If a system initially containing 2 moles of calcium, 3 moles of carbon and 1 mole of calcium carbide is allowed to react to completion, the heat evolved at 298 K will be
30,000 J
60,000 J
90,000 J
2,40,000 J
Surface tension of a liquid
Is due to intermolecular forces of cohesion
Decreases with rise in temperature
Is responsible for the spherical shape of an isolated liquid drop
All (A), (B) & (C)
