PVy = constant, holds good for an isentropic process, which is
Reversible and isothermal
Isothermal and irreversible
Reversible and adiabatic
Adiabatic and irreversible
Correct Answer :
C. Reversible and adiabatic
Related Questions
Which of the following liquid metals has the highest thermal conductivity?
Molten sodium
Molten lead
Mercury
Molten potassium
In the reaction, represented by, 2SO2 + O2 2SO3; ΔH = - 42 kcal; the forward reaction will be favoured by
Low temperature
High pressure
Both (A) and (B)
Neither (A) nor (B)
Efficiency of a heat engine working on Carnot cycle between two temperature levels depends upon the
Two temperatures only
Pressure of working fluid
Mass of the working fluid
Mass and pressure both of the working fluid
Pick out the correct statement.
A real gas on expansion in vacuum gets heated up
An ideal gas on expansion in vacuum gets cooled
An ideal gas on expansion in vacuum gets heated up
A real gas on expansion in vacuum cools down whereas ideal gas remains unaffected
If heat contents of CH4, C2H4 and C3H8 are -17.9, 12.5 and -24.8 kcal/mole respectively, than ΔH for the reaction CH4(g) + C2H4(g) C3H8(g) will be __________ Kcal.
-19.4
-30.2
55.2
-55.2
The adiabatic throttling process of a perfect gas is one of constant enthalpy
In which there is a temperature drop
Which is exemplified by a non-steady flow expansion
Which can be performed in a pipe with a constriction
In which there is an increase in temperature
Enthalpy of a gas depends upon its
Temperature
Mass
Volume
Pressure
The change in __________ is equal to the reversible work for compression in steady state flow process under isothermal condition.
Internal energy
Enthalpy
Gibbs free energy
Helmholtz free energy
Pick out the wrong statement.
The chemical potential of a pure substance depends upon the temperature and pressure
The chemical potential of a component in a system is directly proportional to the escaping tendency of that component
The chemical potential of ith species (μi) in an ideal gas mixture approaches zero as the pressure or mole fraction (xi) tends to be zero at constant temperature
The chemical potential of species 'i' in the mixture (μi) is mathematically represented as,μi = ∂(nG)/∂ni]T,P,nj where, n, ni and nj respectively denote the total number of moles, moles of ith species and all mole numbers except ith species. 'G' is Gibbs molar free energy
A gas has a volume of 27.3 c.c. at 0°C. Its volume at 10°C (if pressure remains unchanged) will be __________ c.c.
2.73
28.3
273
283
Refrigeration capacity of a household refrigerator may be round about __________ tons.
0.15
1.5
4.5
6.5
With increase in reduced temperature, the fugacity co-efficient of a gas at constant reduced pressure
Increases
Decreases
Remain same
Decreases linearly
What happens in a reversible adiabatic expansion process?
Heating takes place
Cooling takes place
Pressure is constant
Temperature is constant
At triple point (for one component system), vapour pressure of solid as compared to that of liquid will be
More
Less
Same
More or less; depending on the system
The gas law (PV = RT) is true for an __________ change.
Isothermal
Adiabatic
Both (A) & (B)
Neither (A) nor (B)
The amount of heat required to decompose a compound into its elements is __________ the heat of formation of that compound from its elements.
Less than
More than
Same as
Not related to
Air enters an adiabatic compressor at 300K. The exit temperature for a compression ratio of 3, assuming air to be an ideal gas (Y = Cp/Cv = 7/5) and the process to be reversible, is
300 × (32/7)
300 × (33/5)
300 × (333/7)
300 × (35/7)
A solute distributes itself between two nonmiscible solvents in contact with each other in such a way that, at a constant temperature, the ratio of its concentrations in two layers is constant, irrespective of its total amount. This is
The distribution law
Followed from Margules equation
A corollary of Henry's law
None of these
First law of thermodynamics is mathematically stated as
dQ = dE + dW
dQ = dE - dW
dE = dQ + dW
dW = dQ + dE
__________ does not change during phase transformation processes like sublimation, melting & vaporisation.
Entropy
Gibbs free energy
Internal energy
All (A), (B) & (C)
In case of a close thermodynamic system, there is __________ across the boundaries.
No heat and mass transfer
No mass transfer but heat transfer
Mass and energy transfer
None of these
Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
Specific __________ does not change during a phase change (e.g. sublimation, melting, vaporisation etc.).
Entropy
Internal energy
Enthalpy
Gibbs free energy
Which one is true for a throttling process?
A gas may have more than one inversion temperatures
The inversion temperature is different for different gases
The inversion temperature is same for all gases
The inversion temperature is the temperature at which Joule-Thomson co-efficient is infinity
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
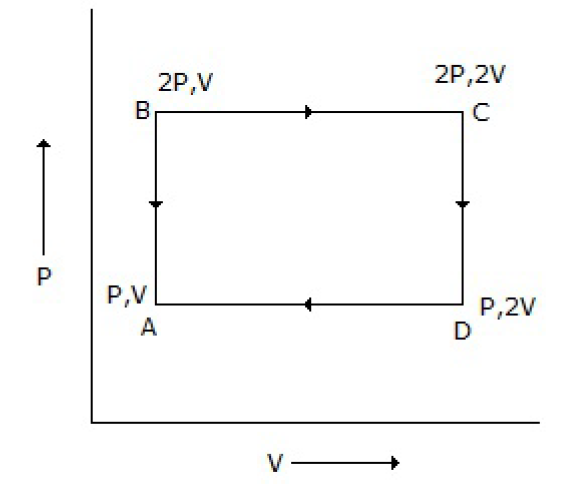
PV
2PV
PV/2
0
The difference between isothermal compressibility and adiabatic compressibility for an ideal gas is
0
+ve
-ve
∞
Which of the following is not an intensive property?
Chemical potential
Surface tension
Heat capacity
None of these
Out of the following refrigeration cycles, which one has the minimum COP (Co-efficient of performance)?
Air cycle
Carnot cycle
Ordinary vapour compression cycle
Vapour compression with a reversible expansion engine
A gas can be liquefied by pressure alone only, when its temperature is __________ its critical temperature.
Less than
More than
Equal to or higher than
Less than or equal to
The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
