Sound waves propagation in air exemplifies an __________ process.
Adiabatic
Isothermal
Isometric
None of these
Correct Answer :
A. Adiabatic
Related Questions
Ideal gas law is applicable at
Low T, low P
High T, high P
Low T, high P
High T, low P
Fugacity is a measure of the
Escaping tendencies of the same substance in different phases of a system
Relative volatility of a mixture of two miscible liquids
Behaviour of ideal gases
None of these
In case of an __________ process, the temperature of the system increases.
Isothermal compression
Isothermal expansion
Adiabatic expansion
Adiabatic compression
The compressibility factor for an ideal gas is 1. Its value for any other real gas is
1
< 1
> 1
Either (B) or (C), depends on the nature of the gas
If the vapour pressure at two temperatures of a solid phase in equilibrium with its liquid phase are known, then the latent heat of fusion can be calculated by the
Maxwell's equation
Clausius-Clapeyron Equation
Van Laar equation
Nernst Heat Theorem
The number of degree of freedom for an Azeotropic mixture of ethanol and water in vapourliquid equilibrium, is
3
1
2
0
Which of the following is not an intensive property?
Molar heat capacity
Internal energy
Viscosity
None of these
Specific/molar Gibbs free energy for a pure substance does not change during
Sublimation
Vaporisation
Melting
Either (A), (B) or (C)
Change of heat content when one mole of compound is burnt in oxygen at constant pressure is called the
Calorific value
Heat of reaction
Heat of combustion
Heat of formation
During adiabatic expansion of gas
Pressure remains constant
Pressure is increased
Temperature remains constant
None of these
The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
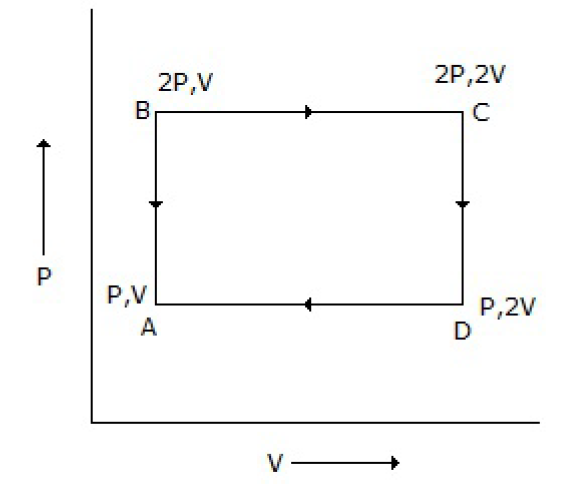
PV
2PV
PV/2
0
Which of the following is not an extensive property?
Free energy
Entropy
Refractive index
None of these
In any spontaneous process,
Only F decreases
Only A decreases
Both F and A decreases
Both F and A increase
Internal energy of an ideal gas
Increases with increase in pressure
Decreases with increase in temperature
Is independent of temperature
None of these
__________ functions are exemplified by heat and work.
Path
Point
State
None of these
Those solutions in which there is no volume change upon mixing the components in the liquid state and which, when diluted do not undergo any heat change (i.e. heat of dilution is zero), are called __________ solutions.
Ideal
Real
Isotonic
None of these
The free energy change for a chemical reaction is given by (where, K = equilibrium constant)
RT ln K
-RT ln K
-R ln K
T ln K
The thermodynamic law, PVy = constant, is not applicable in case of
Ideal compression of air
Free expansion of an ideal gas
Adiabatic expansion of steam in a turbine
Adiabatic compression of a perfect gas
Free energy change at equilibrium is
Zero
Positive
Negative
Indeterminate
In a P-V diagram (for an ideal gas), an isothermal curve will coincide within adiabatic curve (through a point), when
Cp < Cv
Cp = Cv
Cp > Cv
C ≥ Cv
Fugacity and pressure are numerically not equal for the gases
At low temperature and high pressure
At standard state
Both (A) and (B)
In ideal state
An isentropic process is carried out at constant
Volume
Pressure
Temperature
All (A), (B) and (C)
Pick out the wrong statement.
Trouton's ratio of non-polar liquids is calculated using Kistyakowsky equation
Thermal efficiency of a Carnot engine is always less than 1
An equation relating pressure, volume and temperature of a gas is called ideal gas equation
None of these
A gas shows deviation from ideal behaviour at
Low pressure and high temperature
Low pressure and low temperature
Low temperature and high pressure
High temperature and high pressure
Consider the reaction, C + O2 CO2; ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2 → C + O2?
-94 kcal
+94 kcal
> 94 kcal
< -94 kcal
Which of the following is not a common refrigerant?
Freon-12
Ethylene
Ammonia
Carbon dioxide
Gibbs free energy at constant pressure and temperature under equilibrium conditions is
∞
0
Maximum
Minimum
The compressibility factor of a gas is given by (where, V1 = actual volume of the gas V2 = gas volume predicted by ideal gas law)
V1/V2
V2/V1
V1 - V2
V1.V2
The expression for entropy change given by, ΔS = nR ln (V2/V1) + nCv ln (T2/T1) is valid for
Reversible isothermal volume change
Heating of a substance
Cooling of a substance
Simultaneous heating and expansion of an ideal gas
