Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
Correct Answer :
B. Gibbs energy
Related Questions
Specific heat of a gas for a reversible adiabatic process is
Negative
Zero
Infinity
None of these
After throttling, gas temperature
Decreases
Increases
Remain same
May increase or decrease; depends on the nature of the gas
Melting of ice is an example of an __________ process.
Adiabatic
Isothermal
Isometric
None of these
Free energy, fugacity and activity co-efficient are all affected by change in the temperature. The fugacity co-efficient of a gas at constant pressure ____with the increase of reduced temperature.
Decreases
Increases
Remains constant
Decreases logarithmically
Gibbs free energy of a pure fluid approaches __________ as the pressure tends to zero at constant temperature.
Infinity
Minus infinity
Zero
None of these
Third law of thermodynamics is concerned with the
Value of absolute entropy
Energy transfer
Direction of energy transfer
None of these
While dissolving a gas into a liquid at a constant temperature, the ratio of the concentration of the gas in the solution phase and in the gaseous phase is
Infinity
Unity
Constant
Negative
Boiling of liquid is accompanied with increase in the
Vapor pressure
Specific Gibbs free energy
Specific entropy
All (A), (B) and (C)
The freezing point of a liquid decreases when the pressure is increased, if the liquid __________ while freezing.
Contracts
Expands
Does not change in volume
Either (A), (B) or (C)
Enthalpy 'H' is defined as
H = E - PV
H = F - TS
H - E = PV
None of these
The internal energy of an ideal gas does not change in a reversible __________ process.
Isothermal
Adiabatic
Isobaric
Isometric
The efficiency of an Otto engine compared to that of a diesel engine, for the same compression ratio will be
More
Less
Same
Data insufficient to predict
The internal energy of an ideal gas is a function of its __________ only.
Molecular size
Volume
Pressure
Temperature
An isentropic process is carried out at constant
Volume
Pressure
Temperature
All (A), (B) and (C)
Claude gas liquefaction process employs cooling
At constant pressure
By throttling
By expansion in an engine
None of these
Activity co-efficient is a measure of the
Departure from ideal solution behaviour
Departure of gas phase from ideal gas law
Vapour pressure of liquid
None of these
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
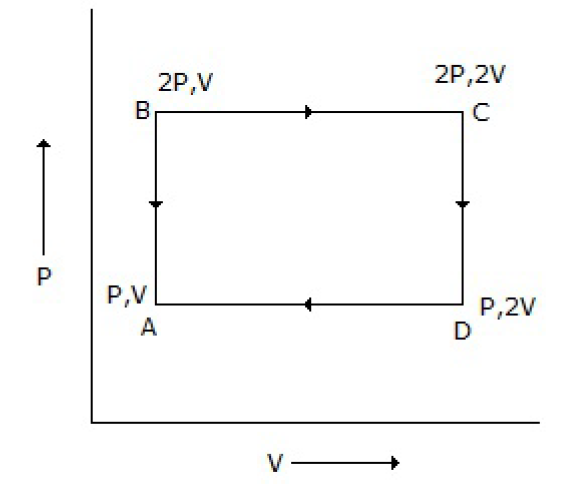
PV
2PV
PV/2
0
Law of corresponding states says that
Two different gases behave similarly, if their reduced properties (i.e. P, V and T) are same
The surface of separation (i. e. the meniscus) between liquid and vapour phase disappears at the critical temperature
No gas can be liquefied above the critical temperature, howsoever high the pressure may be.
The molar heat of energy of gas at constant volume should be nearly constant (about 3 calories)
Chemical potential of ith component of a system is given by
μi = (∂F/∂ni)T, P, ni
μi = (∂A/∂ni)T, P, ni
μi = (∂F/∂ni)T, P
μi = (∂A/∂ni)T, P
Which of the following is not an intensive property?
Chemical potential
Surface tension
Heat capacity
None of these
Pick out the wrong statement.
Activity co-efficient is dimensionless.
In case of an ideal gas, the fugacity is equal to its pressure.
In a mixture of ideal gases, the fugacity of a component is equal to the partial pressure of the component.
The fugacity co-efficient is zero for an ideal gas
Critical temperature is defined as the temperature above which a gas will
Not liquify (barring exceptions)
Immediately liquify
Never liquify however high the pressure may be
None of these
The necessary and sufficient condition for equilibrium between two phases is
The concentration of each component should be same in the two phases
The temperature of each phase should be same
The pressure should be same in the two phases
The chemical potential of each component should be same in the two phases
Heat is added at constant temperature in an ideal __________ cycle.
Stirling
Brayton
Rankine
None of these
If an ideal solution is formed by mixing two pure liquids in any proportion, then the __________ of mixing is zero
Enthalpy
Volume
Both 'a' & 'b'
Neither 'a' nor 'b'
There is a change in __________ during the phase transition.
Volume
Pressure
Temperature
All a, b & c
Degree of freedom of a system consisting of a gaseous mixture of H2 and NH3 will be
0
1
2
3
Clausius-Clapeyron equation is applicable to __________ equilibrium processes.
Solid-vapor
Solid-liquid
Liquid-vapor
All (A), (B) and (C)
The unit of equilibrium constant of a chemical reaction is the same as that of
Molar concentration
Temperature
Internal energy
None of these
In the reaction, represented by, 2SO2 + O2 2SO3; ΔH = - 42 kcal; the forward reaction will be favoured by
Low temperature
High pressure
Both (A) and (B)
Neither (A) nor (B)
