Stirling cycle consists of
Two constant volume and two isentropic processes
Two constant volume and two isothermal processes
Two constant pressure and two isothermal processes
One constant volume, one constant pressure and two isentropic processes
Correct Answer :
B. Two constant volume and two isothermal processes
Related Questions
When a system changes its state from one equilibrium state to another equilibrium state, then the path of successive states through which the system has passed, is known as
Thermodynamic law
Thermodynamic process
Thermodynamic cycle
None of these
The intensity of stress which causes unit strain is called
Unit mass
Modulus of rigidity
Bulk modulus
Modulus of Elasticity
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
In a free expansion process,
W1 - 2 = 0
Q1 - 2 = 0
dU = 0
All of these
In the below figure, the stress corresponding to point D is
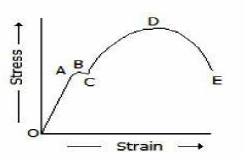
Yield point stress
Breaking stress
Ultimate stress
Elastic limit
The gas constant (R) is equal to the __________ of two specific heats.
Sum
Difference
Product
Ratio
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
When a bar is subjected to a change of temperature and its deformation is prevented, the stress induced in the bar is
Tensile stress
Compressive stress
Shear stress
Thermal stress
The variables which control the physical properties of a perfect gas are
Pressure exerted by the gas
Volume occupied by the gas
Temperature of the gas
All of these
The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
The stress developed in a material at breaking point in extension is called
Breaking stress
Fracture stress
Yield point stress
Ultimate tensile stress
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
Which of the following process can be made reversible with the help of a regenerator?
Constant pressure process
Constant volume process
Constant pvn process
All of these
The amount of heat required to raise the temperature of the unit mass of gas through one degree at constant volume, is called
Specific heat at constant volume
Specific heat at constant pressure
Kilo Joule
None of these
Which of the following cycles is not a reversible cycle?
Carnot
Ericsson
Stirling
None of the above
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
The specific heat of water is
1.817
2512
4.187
None of these
When the gas is heated at constant volume, the heat supplied
Increases the internal energy of the gas and increases the temperature of the gas
Does some external work during expansion
Both (A) and (B)
None of these
The maximum tangential stress in a thick cylindrical shell is always _________ the internal pressure acting on the shell.
Equal to
Less than
Greater than
None of these
The heat supplied to the gas at constant volume is (where m = Mass of gas, cv = Specific heat at constant volume, cp = Specific heat at constant pressure, T2 - T1 = Rise in temperature, and R = Gas constant)
mR (T2 - T1)
mcv (T2 - T1)
mcp (T2 - T1)
mcp (T2 + T1)
The behaviour of super-heated vapour is similar to that of
Perfect gas
Air
Steam
Ordinary gas
When cut-off ratio is __________ the efficiency of Diesel cycle approaches to Otto cycle efficiency.
Zero
1/5
4/5
1
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
In a steady flow process, the ratio of
Heat transfer is constant
Work transfer is constant
Mass flow at inlet and outlet is same
All of these
One molecule of oxygen consists of __________ atoms of oxygen.
2
4
8
16
One reversible heat engine operates between 1600 K and T2 K and another reversible heat engine operates between T2 K and 400 K. If both the engines have the same heat input and output, then temperature T2 is equal to
800 K
1000 K
1200 K
1400 K
Producer gas is obtained by
Partial combustion of coal, coke, anthracite coal or charcoal in a mixed air steam blast
Carbonisation of bituminous coal
Passing steam over incandescent coke
Passing air and a large amount of steam over waste coal at about 650°C
When a gas is heated at constant volume
Its temperature will increase
Its pressure will increase
Both temperature and pressure will increase
Neither temperature nor pressure will increase
Slenderness of a column is zero when
Ends are firmly fixed
Column is supported on all sides throughout the length
Length is equal to radius of gyration
Length is twice the radius of gyration
