The absolute zero pressure can be attained at a temperature of
0°C
273°C
273 K
None of these
Correct Answer :
D. None of these
Related Questions
A coil is cut into two halves, the stiffness of cut coil will be
Double
Half
Same
None of these
The efficiency of Diesel cycle with decrease in cut-off
Increases
Decreases
First increases and then decreases
First decreases and then increases
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
The materials which exhibit the same elastic properties in all directions are called
Homogeneous
Inelastic
Isotropic
Isentropic
A concentrated load is one which
Acts at a point on a beam
Spreads non-uniformly over the whole length of a beam
Spreads uniformly over the whole length of a beam
Varies uniformly over the whole length of a beam
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
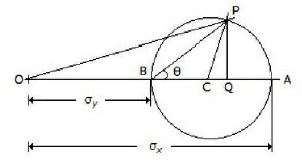
OC
OP
OQ
PQ
In an isothermal process,
There is no change in temperature
There is no change in enthalpy
There is no change in internal energy
All of these
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
The universal gas constant (or molar constant) of a gas is the product of
Molecular mass of the gas and the gas constant
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the specific heat at constant pressure
Molecular mass of the gas and the specific heat at constant volume
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
An isothermal process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
In S. I. units, the value of the universal gas constant is
8.314 J/kg mole-K
83.14 J/kgmole-K
831.4 J/kgmole-K
8314 J/kgmole-K
The strain energy stored in a body due to suddenly applied load compared to when it is applied gradually is
Same
Twice
Four times
Eight times
In an ideal gas turbine plant, it is assumed that the compression and expansion processes are
Isothermal
Isentropic
Polytropic
None of these
If a part is constrained to move and heated, it will develop
Principal stress
Tensile stress
Compressive stress
Shear stress
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
A beam is loaded as cantilever. If the load at the end is increased, the failure will occur
In the middle
At the tip below the load
At the support
Anywhere
When cut-off ratio is __________ the efficiency of Diesel cycle approaches to Otto cycle efficiency.
Zero
1/5
4/5
1
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then maximum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
A molecule consisting of one atom is known as
Mono-atomic
Di-atomic
Tri-atomic
Poly-atomic
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel, the process is known as __________ of fuel.
Atomisation
Carbonisation
Combustion
None of these
The temperature at which the volume of a gas becomes zero is called
Absolute scale of temperature
Absolute zero temperature
Absolute temperature
None of these
When the gas is cooled at constant pressure,
Its temperature increases but volume decreases
Its volume increases but temperature decreases
Both temperature and volume increases
Both temperature and volume decreases
The property of a working substance which increases or decreases as the heat is supplied or removed in a reversible manner, is known as
Enthalpy
Internal energy
Entropy
External energy
Otto cycle is also known as
Constant pressure cycle
Constant volume cycle
Constant temperature cycle
Constant temperature and pressure cycle
Steam coal is a
Pulverised coal
Brown coal
Coking bituminous coal
Non-coking bituminous coal
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
