The buckling load for a given material depends on
Slenderness ratio and area of cross-section
Poisson's ratio and modulus of elasticity
Slenderness ratio and modulus of elasticity
Slenderness ratio, area of cross-section and modulus of elasticity
Correct Answer :
D. Slenderness ratio, area of cross-section and modulus of elasticity
Related Questions
The cycle in which heat is supplied at constant volume and rejected at constant pressure is known as
Dual combustion cycle
Diesel cycle
Atkinson cycle
Rankine cycle
The heat and mechanical energies are mutually convertible. This statement was established by
Boyle
Charles
Joule
None of these
The ultimate tensile stress of mild steel compared to ultimate compressive stress is
Same
More
Less
Unpredictable
One kg of carbon produces __________ kg of carbon dioxide.
3/7
7/3
11/3
3/11
In a tensile test, near the elastic limit zone, the
Tensile strain increases more quickly
Tensile strain decreases more quickly
Tensile strain increases in proportion to the stress
Tensile strain decreases in proportion to the stress
The deformation of a bar under its own weight compared to the deformation of same body subjected to a direct load equal to weight of the body is
Same
Double
Half
Four times
The tensile strength of the welded joint for double fillet is (where s = Leg or size of the weld, l = Length of weld, and σt = Allowable tensile stress for weld metal)
0.5 s.l.σt
s.l.σt
√2 s.l.σt
2.s.l.σt
The deformation per unit length is called
Tensile stress
Compressive stress
Shear stress
Strain
The value of 1 mm of Hg is equal to
1.333 N/m2
13.33 N/m2
133.3 N/m2
1333 N/m2
A hollow shaft of same cross-section area as compared to a solid shaft transmit
Same torque
Less torque
More torque
Unpredictable
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The value of one bar (in S. I. units) is equal to
1 × 102 N/m2
1 × 103 N/m2
1 × 104 N/m2
1 × 105 N/m2
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
For the constant pressure and heat input, the air standard efficiency of gas power cycle is in the order
Dual cycle, Diesel cycle, Otto cycle
Otto cycle, Diesel cycle, Dual cycle
Dual cycle, Otto cycle, Diesel cycle
Diesel cycle, Otto cycle, Dual cycle
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
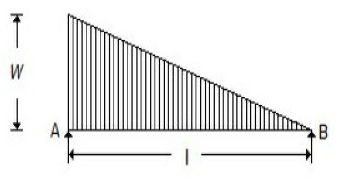
wl/6
wl/3
wl
2wl/3
During which of the following process does heat rejection takes place in Carnot cycle?
Isothermal expansion
Isentropic expansion
Isothermal compression
Isentropic compression
The entropy of water at 0°C is assumed to be
1
0
-1
10
Energy can neither be created nor destroyed, but it can be transformed from one form to another. This statement is known as
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kinetic theory of gases
The limit of eccentricity for no tensile conditions for a column of circular section of diameter (D) is
d/4
d/8
d/12
d/16
The measurement of a thermodynamic property known as temperature is based on
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
None of these
The principal constituents of a fuel are
Carbon and hydrogen
Oxygen and hydrogen
Sulphur and oxygen
Sulphur and hydrogen
Resilience is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of the specimen maximum strain
Energy which can be stored in a body
None of the above
Tensile strength of a material is obtained by dividing the maximum load during the test by the
Area at the time of fracture
Original cross-sectional area
Average of (A) and (B)
Minimum area after fracture
A continuous beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported on more than two supports
Extending beyond the supports
The lower layer of the beam as shown in the below figure, will be

In tension
In compression
Neither in tension nor in compression
None of these
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
The maximum tangential stress in a thick cylindrical shell is always _________ the internal pressure acting on the shell.
Equal to
Less than
Greater than
None of these
