Related Questions
The neutral axis of a transverse section of a beam passes through the centre of gravity of the section and is
In the vertical plane
In the horizontal plane
In the same plane in which the beam bends
At right angle to the plane in which the beam bends
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
According to Kelvin-Planck's statement, a perpetual motion of the __________ is impossible.
First kind
Second kind
Third kind
None of these
A pressure vessel is said to be a thick shell, when
It is made of thick sheets
The internal pressure is very high
The ratio of wall thickness of the vessel to its diameter is less than 1/10.
The ratio of wall thickness of the vessel to its diameter is greater than 1/10.
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
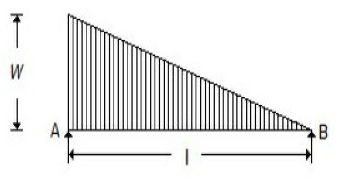
wl/6
wl/3
wl
2wl/3
The amount of heat required to raise the temperature of __________ water through one degree is called kilojoules.
1 g
10 g
100 g
1000 g
If Th is the torque resisting capacity of a hollow shaft and Ts is that of a solid shaft, of the same material, length and weight. Then,
Th > Ts
Th < Ts
Th = Ts
None of these
According to kinetic theory of gases, the velocity of molecules __________ with the increase in temperature.
Remains constant
Increases
Decreases
None of these
The bending moment of a cantilever beam of length l and carrying a uniformly distributed load of w per unit length is __________ at the fixed end.
wl/4
wl/2
wl
wl²/2
The atomic mass of oxygen is
12
14
16
32
Otto cycle efficiency is higher than Diesel cycle efficiency for the same compression ratio and heat input because in Otto cycle
Combustion is at constant volume
Expansion and compression are isentropic
Maximum temperature is higher
Heat rejection is lower
A shaft revolving at ω rad/s transmits torque (T) in Nm. The power developed is
T.ω watts
2π. T.ω watts
2π. T.ω/75 watts
2π. T.ω/4500 watts
The isothermal and adiabatic processes are regarded as
Reversible process
Irreversible process
Reversible or irreversible process
None of these
The efficiency of Diesel cycle with decrease in cut-off
Increases
Decreases
First increases and then decreases
First decreases and then increases
The efficiency of the Carnot cycle may be increased by
Increasing the highest temperature
Decreasing the highest temperature
Increasing the lowest temperature
Keeping the lowest temperature constant
In case of an under-reinforced beam, the depth of actual neutral axis is __________ that of the critical neutral axis.
Same as
Less than
Greater than
None of these
Producer gas is obtained by
Partial combustion of coal, coke, anthracite coal or charcoal in a mixed air steam blast
Carbonisation of bituminous coal
Passing steam over incandescent coke
Passing air and a large amount of steam over waste coal at about 650°C
Which of the following statement is incorrect?
The liquid fuels have higher calorific value than solid fuels
The solid fuels have higher calorific value than liquid fuels
A good fuel should have low ignition point
The liquid fuels consist of hydrocarbons
One kg of ethylene (C2H4) requires 2 kg of oxygen and produces 22/7 kg of carbon dioxide and __________ kg of water or steam.
9/7
11/7
7/4
1/4
When gas is heated at constant pressure, the heat supplied is utilised in
Increasing the internal energy of gas
Doing some external work
Increasing the internal energy of gas and also for doing some external work
None of the above
The amount of heat generated per kg of fuel is known as
Calorific value
Heat energy
Lower calorific value
Higher calorific value
The efficiency of Carnot cycle depends upon
Temperature limits
Pressure ratio
Volume compression ratio
Cut-off ratio and compression ratio
Slenderness of a column is zero when
Ends are firmly fixed
Column is supported on all sides throughout the length
Length is equal to radius of gyration
Length is twice the radius of gyration
Modular ratio of two materials is the ratio of
Strains
Stress and strain
Shear stress and shear strain
Moduli and elasticity
Which of the following is the extensive property of a thermodynamic system?
Pressure
Volume
Temperature
Density
The root mean square velocity of the gas molecules is given by (where k = Boltzmann's constant, T = Absolute temperature, and m = Mass of one molecule of a gas)
√(KT/m)
√(2KT/m)
√(3KT/m)
√(5KT/m)
The gas constant (R) is equal to the __________ of two specific heats.
Sum
Difference
Product
Ratio
The distance between the centres of the rivets in adjacent rows of zigzag riveted joint is known as
Pitch
Back pitch
Diagonal pitch
Diametric pitch
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
Diesel cycle consists of following four processes
Two isothermal and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
