The compression ratio for petrol engines is
3 to 6
5 to 8
15 to 20
20 to 30
Correct Answer :
B. 5 to 8
Related Questions
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
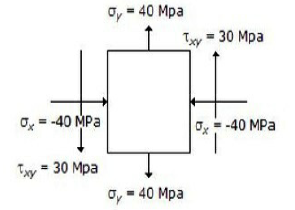
10 MPa
30 MPa
50 MPa
100 MPa
The value of modulus of elasticity for mild steel is of the order of
2.1 × 10⁵ kg/cm²
2.1 × 10⁶ kg/cm²
2.1 × 10⁷ kg/cm²
0.1 × 10⁶ kg/cm²
The oxygen atom is ________ times heavier than the hydrogen atom.
2
8
16
32
The neutral axis of a transverse section of a beam passes through the centre of gravity of the section and is
In the vertical plane
In the horizontal plane
In the same plane in which the beam bends
At right angle to the plane in which the beam bends
The sum of internal energy (U) and the product of pressure and volume (p.v) is known as
Workdone
Entropy
Enthalpy
None of these
The radius of the Mohrs circle in the given figure is equal to
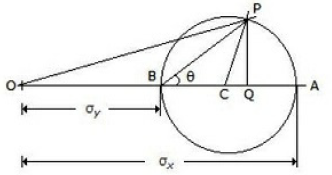
Sum of two principal stresses
Difference of two principal stresses
Half the sum of two principal stresses
Half the difference of two principal stresses
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
Otto cycle consists of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then minimum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
If the slenderness ratio for a column is 100, then it is said to be a _________ column.
Long
Medium
Short
None of these
A pressure vessel is said to be a thick shell, when
It is made of thick sheets
The internal pressure is very high
The ratio of wall thickness of the vessel to its diameter is less than 1/10.
The ratio of wall thickness of the vessel to its diameter is greater than 1/10.
The columns whose slenderness ratio is less than 80, are known as
Short columns
Long columns
Weak columns
Medium columns
The relation between Young's modulus (E), shear modulus (C) and bulk modulus (K) is given by
E = 3K.C/(3K + C)
E = 6K.C/(3K + C)
E = 9K.C/(3K + C)
E = 12K.C/(3K + C)
One reversible heat engine operates between 1600 K and T2 K and another reversible heat engine operates between T2 K and 400 K. If both the engines have the same heat input and output, then temperature T2 is equal to
800 K
1000 K
1200 K
1400 K
A concentrated load is one which
Acts at a point on a beam
Spreads non-uniformly over the whole length of a beam
Spreads uniformly over the whole length of a beam
Varies uniformly over the whole length of a beam
A double strap butt joint with equal straps is
Always in single shear
Always in double shear
Either in single shear or double shear
None of these
Reversed joule cycle is called
Carnot cycle
Rankine cycle
Brayton cycle
Bell Coleman cycle
Which of the following is the correct statement of the second law of thermodynamics?
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work.
It is impossible to transfer heat from a body at a lower temperature to a higher temperature, without the aid of an external source.
There is a definite amount of mechanical energy, which can be obtained from a given quantity of heat energy.
All of the above
The measurement of a thermodynamic property known as temperature is based on
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
None of these
A column of length (l) with both ends fixed may be considered as equivalent to a column of length __________ with both ends hinged.
l/8
l/4
l/2
l
The principal constituents of a fuel are
Carbon and hydrogen
Oxygen and hydrogen
Sulphur and oxygen
Sulphur and hydrogen
Which of the following is the extensive property of a thermodynamic system?
Pressure
Volume
Temperature
Density
The value of shear stress which is induced in the shaft due to the applied couple varies
From maximum at the centre to zero at the circumference
From zero at the centre to maximum at the circumference
From maximum at the centre to minimum at the circumference
From minimum at the centre to maximum at the circumference
Which of the following is correct?
Absolute pressure = Gauge pressure + Atmospheric pressure
Gauge pressure = Absolute pressure + Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
The property of a material by virtue of which a body returns to its original, shape after removal of the load is called
Plasticity
Elasticity
Ductility
Malleability
A cycle consisting of two isothermal and two isentropic processes, is known as
Carnot cycle
Stirling cycle
Ericsson cycle
Joule cycle
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
When a body is subjected to three mutually perpendicular stresses, of equal intensity, the ratio of direct stress to the corresponding volumetric strain is known as
Young's modulus
Modulus of rigidity
Bulk modulus
Poisson's ratio
When a gas is heated at constant pressure
Its temperature will increase
Its volume will increase
Both temperature and volume will increase
Neither temperature not volume will increase
The section modulus of a circular section about an axis through its C.G., is
πd²/4
πd²/16
πd3/16
πd3/32
