The deformation of a bar under its own weight compared to the deformation of same body subjected to a direct load equal to weight of the body is
Same
Double
Half
Four times
Correct Answer :
C. Half
Related Questions
Stirling and Ericsson cycles are
Reversible cycles
Irreversible cycles
Semi-reversible cycles
Quasi-static cycles
When the gas is heated at constant volume, the heat supplied
Increases the internal energy of the gas and increases the temperature of the gas
Does some external work during expansion
Both (A) and (B)
None of these
The heating of a gas at constant pressure is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
Diamond riveted joint can be adopted in the case of following type of joint
Butt joint
Lap joint
Double riveted lap joints
All types of joints
The behaviour of a perfect gas, undergoing any change in the variables which control physical properties, is governed by
Boyle's law
Charles' law
Gay-Lussac law
All of these
The ratio of specific heat at constant pressure (cp) and specific heat at constant volume (cv) is always __________ one.
Equal to
Less than
Greater than
None of these
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
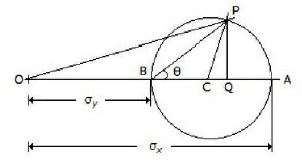
OC
OP
OQ
PQ
One kg of carbon monoxide requires __________ kg of oxygen to produce 11/7 kg of carbon dioxide gas.
4/7
11/4
9/7
All of these
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
The main cause for the irreversibility is
Mechanical and fluid friction
Unrestricted expansion
Heat transfer with a finite temperature difference
All of the above
The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
A cantilever beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported at its ends
Supported on more than two supports
The thermal efficiency of an ideal gas turbine plant is given by (where r = Pressure ratio)
rγ - 1
1 - rγ - 1
1 - (1/r) γ/γ - 1
1 - (1/r) γ - 1/ γ
The general gas energy equation is (where Q1 - 2 = Heat supplied, dU = Change in internal energy, and W1 - 2 = Work done in heat units)
Q1 - 2 = dU + W1 - 2
Q1 - 2 = dU - W1 - 2
Q1 - 2 = dU/W1 - 2
Q1 - 2 = dU × W1 - 2
True stress strain-curve for materials is plotted between
Load/original cross-sectional area and change in length/original length
Load/ instantaneous cross-sectional area and loge (original area/ instantaneous area)
Load/ instantaneous cross-sectional area and change in length/ original length
Load/ instantaneous area and instantaneous area/original area
According to kinetic theory of gases, the velocity of molecules __________ with the increase in temperature.
Remains constant
Increases
Decreases
None of these
The absolute zero temperature is taken as
-273°C
73°C
237°C
-237°C
The shape of cantilever for uniformly distributed load will be
Straight line
Parabolic
Elliptical
Cubic
The measurement of a thermodynamic property known as temperature is based on
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
None of these
Which of the following statement is correct?
The heat and work are boundary phenomena
The heat and work represent the energy crossing the boundary of the system
The heat and work are path functions
All of the above
If a part is constrained to move and heated, it will develop
Principal stress
Tensile stress
Compressive stress
Shear stress
Diesel cycle consists of __________ processes.
Two constant volume and two isentropic
Two constant pressure and two isentropic
Two constant volume and two isothermal
One constant pressure, one constant volume and two isentropic
An open system is one in which
Heat and work crosses the boundary of the system, but the mass of the working substance does not crosses the boundary of the system
Mass of the working substance crosses the boundary of the system but the heat and work does not crosses the boundary of the system
Both the heat and work as well as mass of the working substance crosses the boundary of the system
Neither the heat and work nor the mass of the working substance crosses the boundary of the system
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The rivets are used for __________ fastenings.
Permanent
Temporary
Semi-permanent
None of these
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
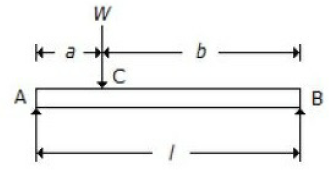
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
Steam coal is a
Pulverised coal
Brown coal
Coking bituminous coal
Non-coking bituminous coal
When a rectangular beam is loaded transversely, the maximum compressive stress is developed on the
Top layer
Bottom layer
Neutral axis
Every cross-section
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
