The deformation per unit length is called
Tensile stress
Compressive stress
Shear stress
Strain
Correct Answer :
D. Strain
Related Questions
In the torsion equation T/J = τ/r = Gθ/ L, the term J/R is called
Shear modulus
Section modulus
Polar modulus
None of these
The value of cp/cv for air is
1
1.4
1.45
2.3
The distance between the centres of the rivets in adjacent rows of zigzag riveted joint is known as
Pitch
Back pitch
Diagonal pitch
Diametric pitch
According to Avogadro's law, the density of any two gases is __________ their molecular masses, if the gases are at the same temperature and pressure.
Equal to
Directly proportional to
Inversely proportional to
None of these
A cylindrical section having no joint is known as
Joint less section
Homogeneous section
Perfect section
Seamless section
For a beam, as shown in the below figure, when the load W is applied in the centre of the beam, the maximum deflection is
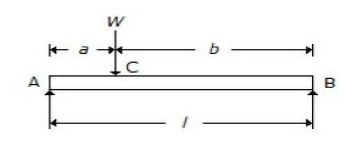
Wl3 / 48EI
5Wl3 / 384EI
Wl3 / 392EI
Wl3 / 384EI
The isothermal and adiabatic processes are regarded as
Reversible process
Irreversible process
Reversible or irreversible process
None of these
The behaviour of super-heated vapour is similar to that of
Perfect gas
Air
Steam
Ordinary gas
All perfect gases change in volume by 1/273th of its original volume at 0°C for every 1°C change in temperature, when the pressure remains constant. This statement is called
Boyle's law
Charles' law
Gay-Lussac law
Joule's law
In order to prevent crushing of masonry at the base of the dam, the maximum stress should be __________ the permissible stress of the soil.
Equal to
Less than
More than
None of these
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
For a simply supported beam of length l, when a concentrated load W is applied in the centre of the beam, the maximum deflection is
5WL³/ 384EI
WL³/384EI
WL³/ 348EI
WL³/ 48EI
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
One kg of carbon monoxide requires 4/7 kg of oxygen and produces
11/3 kg of carbon dioxide gas
7/3 kg of carbon monoxide gas
11/7 kg of carbon dioxide gas
8/3 kg of carbon monoxide gas
A composite bar made up of steel and copper bars of equal lengths are heated through 100°C. The stresses developed shall be
Tensile in both the material
Tensile in steel and compressive in copper
Compressive in steel and tensile in copper
Compressive in both the materials
In a prismatic member made of two materials so joined that they deform equally under axial stress, the unit stresses in two materials are
Equal
Proportional to their respective moduli of elasticity
Inversely proportional to their moduli of elasticity
Average of the sum of moduli of elasticity
Energy can neither be created nor destroyed, but it can be transformed from one form to another. This statement is known as
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kinetic theory of gases
Carnot cycle consists of
Two constant volume and two isentropic processes
Two isothermal and two isentropic processes
Two constant pressure and two isentropic processes
One constant volume, one constant pressure and two isentropic processes
For the constant pressure and heat input, the air standard efficiency of gas power cycle is in the order
Dual cycle, Diesel cycle, Otto cycle
Otto cycle, Diesel cycle, Dual cycle
Dual cycle, Otto cycle, Diesel cycle
Diesel cycle, Otto cycle, Dual cycle
The following cycle is used for air craft refrigeration
Brayton cycle
Joule cycle
Carnot cycle
Reversed Brayton cycle
The universal gas constant (or molar constant) of a gas is the product of
Molecular mass of the gas and the gas constant
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the specific heat at constant pressure
Molecular mass of the gas and the specific heat at constant volume
When a body is subjected to a direct tensile stress (σ) in one plane, then maximum normal stress occurs at a section inclined at __________ to the normal of the section.
0°
30°
45°
90°
In open cycle gas turbine plants
The indirect heat exchanger and cooler is avoided
Direct combustion system is used
A condenser is used
All of the above
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
The efficiency of the Carnot cycle is (where T1 and T2 = Highest and lowest temperature during the cycle)
(T1/T2) - 1
1 - (T1/T2)
1 - (T2/T1)
1 + (T2/T1)
When a body is subjected to three mutually perpendicular stresses, of equal intensity, the ratio of direct stress to the corresponding volumetric strain is known as
Young's modulus
Modulus of rigidity
Bulk modulus
Poisson's ratio
In case of an under-reinforced beam, the depth of actual neutral axis is __________ that of the critical neutral axis.
Same as
Less than
Greater than
None of these
Steam coal is a
Pulverised coal
Brown coal
Coking bituminous coal
Non-coking bituminous coal
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
The ratio of direct stress to volumetric strain in case of a body subjected to three mutually perpendicular stresses of equal intensity, is equal to
Young's modulus
Bulk modulus
Modulus of rigidity
Modulus of elasticity
