The efficiency of Diesel cycle with decrease in cut-off
Increases
Decreases
First increases and then decreases
First decreases and then increases
Correct Answer :
A. Increases
Related Questions
The radius of the Mohrs circle in the given figure is equal to
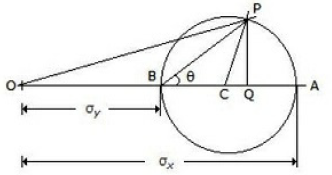
Sum of two principal stresses
Difference of two principal stresses
Half the sum of two principal stresses
Half the difference of two principal stresses
When a rectangular beam is loaded transversely, the maximum compressive stress is developed on the
Top layer
Bottom layer
Neutral axis
Every cross-section
Select the correct statement as per Charles' law
p.v = constant, if T is kept constant
v/T = constant, if p is kept constant
p/T = constant, if v is kept constant
T/p = constant, if v is kept constant
When coal is first dried and then crushed to a fine powder by pulverising machine, the resulting fuel is called
Wood charcoal
Bituminous coal
Briquetted coal
None of these
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
A process of heating crude oil to a high temperature under a very high pressure to increase the yield of lighter distillates, is known as
Cracking
Carbonisation
Fractional distillation
Full distillation
In a tensile test, near the elastic limit zone, the
Tensile strain increases more quickly
Tensile strain decreases more quickly
Tensile strain increases in proportion to the stress
Tensile strain decreases in proportion to the stress
True stress strain-curve for materials is plotted between
Load/original cross-sectional area and change in length/original length
Load/ instantaneous cross-sectional area and loge (original area/ instantaneous area)
Load/ instantaneous cross-sectional area and change in length/ original length
Load/ instantaneous area and instantaneous area/original area
An open system is one in which
Heat and work crosses the boundary of the system, but the mass of the working substance does not crosses the boundary of the system
Mass of the working substance crosses the boundary of the system but the heat and work does not crosses the boundary of the system
Both the heat and work as well as mass of the working substance crosses the boundary of the system
Neither the heat and work nor the mass of the working substance crosses the boundary of the system
The efficiency of Diesel cycle with decrease in cut-off
Increases
Decreases
First increases and then decreases
First decreases and then increases
A process, in which the working substance neither receives nor gives out heat to its surroundings during its expansion or compression, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
The value of specific heat at constant pressure (cp) is __________ that of at constant volume (cv).
Less than
Equal to
More than
None of these
Which of the following is an intensive property of a thermodynamic system?
Volume
Temperature
Mass
Energy
The natural solid fuel is
Wood
Coke
Anthracite coal
Pulverised coal
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
According to Avogadro's law, the density of any two gases is __________ their molecular masses, if the gases are at the same temperature and pressure.
Equal to
Directly proportional to
Inversely proportional to
None of these
The cycle in which heat is supplied at constant volume and rejected at constant pressure is known as
Dual combustion cycle
Diesel cycle
Atkinson cycle
Rankine cycle
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
The fuel mostly used in cement industry and in metallurgical processes is
Wood charcoal
Bituminous coke
Pulverised coal
Coke
In order to know whether a column is long or short, we must know its
Ultimate shear stress of the column
Factor of safety
Torque resisting capacity
Slenderness ratio
In the torsion equation T/J = τ/r = Gθ/ L, the term J/R is called
Shear modulus
Section modulus
Polar modulus
None of these
The energy stored in a body when strained within elastic limit is known as
Resilience
Proof resilience
Strain energy
Impact energy
A material obeys hooks law up to
Plastic limit
Elastic limit
Yield point
Limit of proportionality
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
A mixture of gas expands from 0.03 m3 to 0.06 m3 at a constant pressure of 1 MPa and absorbs 84 kJ of heat during the process. The change in internal energy of the mixture is
30 kJ
54 kJ
84 kJ
114 kJ
According to Avogadro's law
The product of the gas constant and the molecular mass of an ideal gas is constant
The sum of partial pressure of the mixture of two gases is sum of the two
Equal volumes of all gases, at the same temperature and pressure, contain equal number of molecules
All of the above
The neutral axis of the cross-section a beam is that axis at which the bending stress is
Zero
Minimum
Maximum
Infinity
The principal constituents of a fuel are
Carbon and hydrogen
Oxygen and hydrogen
Sulphur and oxygen
Sulphur and hydrogen
Resilience is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of the specimen maximum strain
Energy which can be stored in a body
None of the above
The pull required to tear off the plate per pitch length is (where p = Pitch of rivets, t = Thickness of plates, and σt, τ and σc = Permissible tensile, shearing and crushing stresses respectively)
(p - 2d) t × σc
(p - d) t × τ
(p - d) t × σt
(2p - d) t × σt
