The efficiency of the Carnot cycle may be increased by
Increasing the highest temperature
Decreasing the highest temperature
Increasing the lowest temperature
Keeping the lowest temperature constant
Correct Answer :
A. Increasing the highest temperature
Related Questions
Which of the following is correct?
Gauge pressure = Absolute pressure + Atmospheric pressure
Absolute pressure = Gauge pressure + Atmospheric pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
The strain energy stored in a solid circular shaft subjected to shear stress (τ), is: (Where G = Modulus of rigidity for the shaft material)
τ²/ 2G × Volume of shaft
τ/ 2G × Volume of shaft
τ²/ 4G × Volume of shaft
τ/ 4G × Volume of shaft
The measurement of a thermodynamic property known as temperature is based on
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
None of these
When wood is heated with a limited supply of air to a temperature not less than 280°C, the resulting fuel is
Coke
Wood charcoal
Bituminous coal
Briquetted coal
The efficiency of a gas turbine is given by
(Net work output)/(Workdone by the turbine)
(Net work output)/(Heat supplied)
(Actual temperature drop)/(Isentropic temperature drop)
(Isentropic increase in temperature)/(Actual increase in temperature)
Otto cycle consists of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
The thermal efficiency of an ideal gas turbine plant is given by (where r = Pressure ratio)
rγ - 1
1 - rγ - 1
1 - (1/r) γ/γ - 1
1 - (1/r) γ - 1/ γ
A material obeys hooks law up to
Plastic limit
Elastic limit
Yield point
Limit of proportionality
A composite shaft consisting of two stepped portions having spring constants K₁ and K₂ is held between two rigid supports at the ends. Its equivalent spring constant is
K₁ K₂
(K₁ + K₂)/ 2
(K₁ + K₂)/ K₁ K₂
K₁ K₂/ (K₁ + K₂)
The __________ states that change of internal energy of a perfect gas is directly proportional to the change of temperature.
Boyle's law
Charle's law
Gay-Lussac law
Joule's law
The unit of energy is S. I. units is
Joule (J)
Joule metre (Jm)
Watt (W)
Joule/metre (J/m)
The ideal efficiency of a Brayton cycle with regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
The gas constant (R) is equal to the
Sum of two specific heats
Difference of two specific heats
Product of two specific heats
Ratio of two specific heats
When the gas is heated at constant volume, the heat supplied
Increases the internal energy of the gas and increases the temperature of the gas
Does some external work during expansion
Both (A) and (B)
None of these
The property of a material which allows it to be drawn into a smaller section is called
Plasticity
Ductility
Elasticity
Malleability
Proof resilience per material is known as
Resilience
Proof resilience
Modulus of resilience
Toughness
A circular shaft fixed at, A has diameter D for half of its length and diameter D/2 over the other half, as shown in the below figure. If the rotation of B relative to A is 0.1 radian, the rotation of C relative to B will be
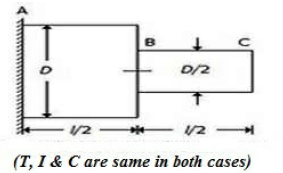
0.4 radian
0.8 radian
1.6 radian
3.2 radian
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
When a gas is heated at constant volume
Its temperature will increase
Its pressure will increase
Both temperature and pressure will increase
Neither temperature nor pressure will increase
When both ends of a column are fixed, the effective length is
Its own length
Twice its length
Half its length
1/√2 × its length
A boiler shell 200 cm diameter and plate thickness 1.5 cm is subjected to internal pressure of 1.5 MN/m, and then the hoop stress will be
30 MN/m²
50 MN/m²
100 MN/m²
200 MN/m²
Formula based on IS codes is based on
Straight line formula
Eulers formula
Rankines formula
Secant formula
A column is said to be a short column, when
Its length is very small
Its cross-sectional area is small
The ratio of its length to the least radius of gyration is less than 80
The ratio of its length to the least radius of gyration is more than 80
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
If the slenderness ratio for a column is 100, then it is said to be a _________ column.
Long
Medium
Short
None of these
Which of the following is the extensive property of a thermodynamic system?
Pressure
Volume
Temperature
Density
Otto cycle consists of
Two constant volume and two isentropic processes
Two constant pressure and two isentropic processes
Two constant volume and two isothermal processes
One constant pressure, one constant volume and two isentropic processes
The universal gas constant (or molar constant) of a gas is the product of
Molecular mass of the gas and the gas constant
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the specific heat at constant pressure
Molecular mass of the gas and the specific heat at constant volume
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
