The efficiency of the dual combustion cycle for the same compression ratio is __________ Diesel cycle.
Greater than
Less than
Equal to
None of these
Correct Answer :
A. Greater than
Related Questions
An isothermal process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The value of cp/cv for air is
1
1.4
1.45
2.3
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
One molecule of oxygen consists of __________ atoms of oxygen.
2
4
8
16
For the same compression ratio, the efficiency of dual combustion cycle is
Greater than Diesel cycle and less than Otto cycle
Less than Diesel cycle and greater than Otto cycle
Greater than Diesel cycle
Less than Diesel cycle
The compression ratio for petrol engines is
3 to 6
5 to 8
15 to 20
20 to 30
A cantilever beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported at its ends
Supported on more than two supports
The change of entropy, when heat is absorbed by the gas, is
Positive
Negative
Positive or negative
None of these
Which of the following gas is mostly used in town for street and domestic lighting and heating?
Producer gas
Coal gas
Mond gas
Coke oven gas
A material obeys hooks law up to
Plastic limit
Elastic limit
Yield point
Limit of proportionality
When a body is subjected to biaxial stress i.e. direct stresses (σx) and (σy) in two mutually perpendicular planes accompanied by a simple shear stress (τxy), then maximum normal stress is
(σx + σy)/2 + (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx + σy)/2 - (1/2) × √[(σx - σy)² + 4 τ²xy]
(σx - σy)/2 + (1/2) × √[(σx + σy)² + 4 τ²xy]
(σx - σy)/2 - (1/2) × √[(σx + σy)² + 4 τ²xy]
For the same compression ratio, the efficiency of Diesel cycle is __________ Otto cycle.
Greater than
Less than
Equal to
None of these
Two shafts 'A' and 'B' transmit the same power. The speed of shaft 'A' is 250 r.p.m. and that of shaft 'B' is 300 r.p.m.
The shaft 'B' has the greater diameter
The shaft 'A' has the greater diameter
Both are of same diameter
None of these
A molecule consisting of one atom is known as
Mono-atomic
Di-atomic
Tri-atomic
Poly-atomic
An open cycle gas turbine works on
Carnot cycle
Otto cycle
Joule's cycle
Stirling cycle
The property of a material by virtue of which it can be beaten or rolled into plates is called
Malleability
Ductility
Plasticity
Elasticity
Which of the following is a reversible non-flow process?
Isochoric process
Isobaric process
Hyperbolic process
All of these
When the expansion or compression takes place according to the law pvn = C, the process is known as
Isothermal process
Adiabatic process
Hyperbolic process
Polytropic process
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The atomic mass of sulphur is
12
14
16
32
Modulus of rigidity may be defined as the ratio of
Linear stress to lateral strain
Lateral strain to linear strain
Linear stress to linear strain
Shear stress to shear strain
When a thin cylindrical shell is subjected to an internal pressure, the volumetric strain is (where ε₁ = Hoop strain, and ε₂ = Longitudinal strain)
2ε₁ - ε₂
2ε₁ + ε₂
2ε₂ - ε₁
2ε₂ + ε₁
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
In the below figure, the plastic range occurs
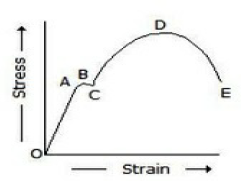
Before point A
Beyond point A
Between points A and D
Between points D and E
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
Proof resilience per material is known as
Resilience
Proof resilience
Modulus of resilience
Toughness
When two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This statement is called
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kelvin Planck's law
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
