The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
Correct Answer :
B. Thermodynamic equation of state
Related Questions
Fugacity co-efficient of a substance is the ratio of its fugacity to
Mole fraction
Activity
Pressure
Activity co-efficient
What is the degree of freedom for two miscible (non-reacting) substances in vapor-liquid equilibrium forming an azeotrope?
0
1
2
3
The number of degrees of freedom for an azeotropic mixture in a two component vapour-liquid equilibria is/are
Zero
One
Two
Three
The adiabatic throttling process of a perfect gas is one of constant enthalpy
In which there is a temperature drop
Which is exemplified by a non-steady flow expansion
Which can be performed in a pipe with a constriction
In which there is an increase in temperature
The internal energy of an ideal gas is a function of its __________ only.
Molecular size
Volume
Pressure
Temperature
If an ideal solution is formed by mixing two pure liquids in any proportion, then the __________ of mixing is zero
Enthalpy
Volume
Both 'a' & 'b'
Neither 'a' nor 'b'
Dry ice is
Moisture free ice
Solid helium
Solid carbon dioxide
None of these
A gas performs the maximum work, when it expands
Non-uniformly
Adiabatically
Isobarically
Isothermally
The unity of Planck's constant 'h' in the equation, E = hv is
J/s
J.S
J/kmol
kmol/J
The first law of thermodynamics is a restatement of the law of conservation of
Mass
Energy
Momentum
None of these
A system undergoes a change from a given initial state to a given final state either by an irreversible process or by a reversible process, then (where, Δ S1 and Δ SR are the entropy changes of the system for the irreversible and reversible processes respectively)
Δ S1 is always < Δ SR
Δ S1 is sometimes > Δ SR
Δ S1 is always > Δ SR
Δ S1 is always = Δ SR
Linde gas liquefaction process employs cooling
By throttling
By expansion in an engine
At constant pressure
None of these
For a constant volume process
dE = CpdT
dE = CvdT
dQ = dE + pdV
dW = pdV
Air-refrigeration cycle
Is the most efficient of all refrigeration cycles
Has very low efficiency
Requires relatively large quantities of air to achieve a significant amount of refrigeration
Both (B) and (C)
The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
In reactions involving solids and liquids (where change in volume is negligible), the heat of reaction at constant pressure as compared to that at constant volume is
More
Less
Same
Unpredictable; depends on the particular reaction
Law of corresponding states says that
Two different gases behave similarly, if their reduced properties (i.e. P, V and T) are same
The surface of separation (i. e. the meniscus) between liquid and vapour phase disappears at the critical temperature
No gas can be liquefied above the critical temperature, howsoever high the pressure may be.
The molar heat of energy of gas at constant volume should be nearly constant (about 3 calories)
Entropy change for an irreversible isolated system is
∞
0
< 0
> 0
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
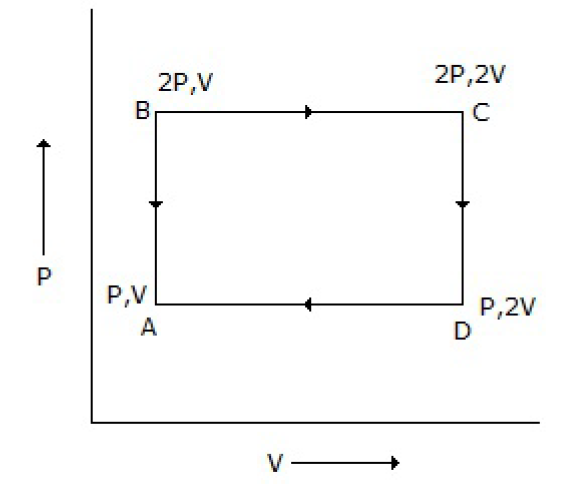
PV
2PV
PV/2
0
Pick out the wrong statement:
The expansion of a gas in vacuum is an irreversible process
An isometric process is a constant pressure process
Entropy change for a reversible adiabatic process is zero
Free energy change for a spontaneous process is negative
Lenz's law results from the law of conservation of
Mass
Momentum
Energy
None of these
Third law of thermodynamics is concerned with the
Value of absolute entropy
Energy transfer
Direction of energy transfer
None of these
Entropy is a measure of the __________ of a system.
Disorder
Orderly behaviour
Temperature changes only
None of these
Heating of water under atmospheric pressure is an __________ process.
Isochoric
Isobaric
Adiabatic
Isothermal
The expression for entropy change given by, ΔS = - nR ln (P2/P1), holds good for
Expansion of a real gas
Reversible isothermal volume change
Heating of an ideal gas
Cooling of a real gas
No work is done by the system, when a reaction occurs at constant
Volume
Temperature
Pressure
None of these
Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
The amount of heat required to decompose a compound into its elements is __________ the heat of formation of that compound from its elements.
Less than
More than
Same as
Not related to
Steam undergoes isentropic expansion in a turbine from 5000 kPa and 400°C (entropy = 6.65 kJ/kg K) to 150 kPa) (entropy of saturated liquid = 1.4336 kJ/kg. K, entropy of saturated vapour = 7.2234 kJ/kg. K) The exit condition of steam is
Superheated vapour
Partially condensed vapour with quality of 0.9
Saturated vapour
Partially condensed vapour with quality of 0.1
Gases are cooled in Joule-Thomson expansion, when it is __________ inversion temperature.
Below
At
Above
Either 'b' or 'c'
