The general gas energy equation is (where Q1 - 2 = Heat supplied, dU = Change in internal energy, and W1 - 2 = Work done in heat units)
Q1 - 2 = dU + W1 - 2
Q1 - 2 = dU - W1 - 2
Q1 - 2 = dU/W1 - 2
Q1 - 2 = dU × W1 - 2
Correct Answer :
A. Q1 - 2 = dU + W1 - 2
Related Questions
In the tensile test, the phenomenon of slow extension of the material, i. e. stress increasing with the time at a constant load is called
Creeping
Yielding
Breaking
Plasticity
A pressure vessel is said to be a thick shell, when
It is made of thick sheets
The internal pressure is very high
The ratio of wall thickness of the vessel to its diameter is less than 1/10.
The ratio of wall thickness of the vessel to its diameter is greater than 1/10.
Which of the following is an intensive property of a thermodynamic system?
Volume
Temperature
Mass
Energy
The throttling process is __________ process.
Reversible
Irreversible
Reversible or irreversible
None of these
One Joule (J) is equal to
1 N-m
1 kN-m
10 N-m/s
10 kN-m/s
Modulus of rigidity is defined as the ratio of
Longitudinal stress to longitudinal strain
Volumetric stress to volumetric strain
Lateral stress to Lateral strain
Shear stress to shear strain
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
Which of the following materials is most elastic?
Rubber
Plastic
Brass
Steel
The mass of carbon per kg of flue gas is given by
(11/3) CO2 + (3/7) CO
(3/7) CO2 + (11/3) CO
(7/3) CO2 + (3/11) CO
(3/11) CO2 + (7/3) CO
The efficiency and work ratio of a simple gas turbine cycle are
Very low
Low
High
Very high
The value of one bar (in S. I. units) is equal to
1 × 102 N/m2
1 × 103 N/m2
1 × 104 N/m2
1 × 105 N/m2
Select the wrong statement
A Joule cycle consists of two constant volume and two isentropic processes.
An Otto cycle consists of two constant volume and two isentropic processes.
An Ericsson cycle consists of two constant pressure and two isothermal processes.
All of the above
Strain re-setters are used to
Measure shear strain
Measure linear strain
Measure volumetric strain
Relieve strain
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
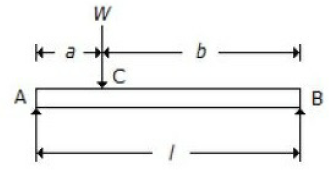
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
Diamond riveted joint can be adopted in the case of following type of joint
Butt joint
Lap joint
Double riveted lap joints
All types of joints
The value of cp/cv for air is
1
1.4
1.45
2.3
The percentage reduction in area of a cast iron specimen during tensile test would be of the order of
More than 50 %
25-50 %
10-25 %
Negligible
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
A circular shaft fixed at, A has diameter D for half of its length and diameter D/2 over the other half, as shown in the below figure. If the rotation of B relative to A is 0.1 radian, the rotation of C relative to B will be
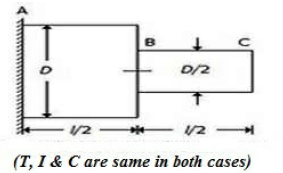
0.4 radian
0.8 radian
1.6 radian
3.2 radian
The value of Poisson's ratio for steel is between
0.01 to 0.1
0.23 to 0.27
0.25 to 0.33
0.4 to 0.6
The ratio of root mean square velocity to average velocity of gas molecules at a particular temperature is
0.086
1.086
1.086
4.086
The extremeties of any diameter on Mohr's circle represent
Principal stresses
Normal stresses on planes at 45°
Shear stresses on planes at 45°
Normal and shear stresses on a plane
The value of modulus of elasticity for mild steel is of the order of
2.1 × 10⁵ kg/cm²
2.1 × 10⁶ kg/cm²
2.1 × 10⁷ kg/cm²
0.1 × 10⁶ kg/cm²
A molecule consisting of one atom is known as
Mono-atomic
Di-atomic
Tri-atomic
Poly-atomic
The general gas equation is (where p = Pressure, v = Volume, m = mass, T = Absolute temperature, and R = Gas constant)
pv = mRT
pv = RTm
pvm = C
pv = (RT)m
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
Diesel cycle consists of following four processes
Two isothermal and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
The intensity of stress which causes unit strain is called
Unit mass
Modulus of rigidity
Bulk modulus
Modulus of Elasticity
The relation between Young's modulus (E), shear modulus (C) and bulk modulus (K) is given by
E = 3K.C/(3K + C)
E = 6K.C/(3K + C)
E = 9K.C/(3K + C)
E = 12K.C/(3K + C)
The efficiency of Diesel cycle approaches to Otto cycle efficiency when
Cut-off is increased
Cut-off is decreased
Cut-off is zero
Cut-off is constant
