The general law of expansion or compression is pvn = C, The process is said to be hyperbolic, if n is equal to
0
1
γ
∝
Correct Answer :
B. 1
Related Questions
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
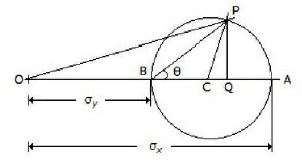
OC
OP
OQ
PQ
When a body is subjected to a direct tensile stress (σx) in one plane accompanied by a simple shear stress (τxy), the maximum normal stress i
(σx/2) + (1/2) × √(σx² + 4 τ²xy)
(σx/2) - (1/2) × √(σx² + 4 τ²xy)
(σx/2) + (1/2) × √(σx² - 4 τ²xy)
(1/2) × √(σx² + 4 τ²xy)
When the gas is cooled at constant pressure,
Its temperature increases but volume decreases
Its volume increases but temperature decreases
Both temperature and volume increases
Both temperature and volume decreases
Strain is defined as the ratio of
Change in volume to original volume
Change in length to original length
Change in cross-sectional area to original cross-sectional area
Any one of the above
Which of the following is an intensive property of a thermodynamic system?
Volume
Temperature
Mass
Energy
A composite shaft consisting of two stepped portions having spring constants K₁ and K₂ is held between two rigid supports at the ends. Its equivalent spring constant is
K₁ K₂
(K₁ + K₂)/ 2
(K₁ + K₂)/ K₁ K₂
K₁ K₂/ (K₁ + K₂)
The stress necessary to initiate yielding is
Considerably greater than that necessary to continue it
Considerably lesser than that necessary to continue it
Greater than that necessary to stop it
Lesser than that necessary to stop it
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
The specific heat of water is
1.817
2512
4.187
None of these
The maximum bending moment for the beam shown in the below figure, is
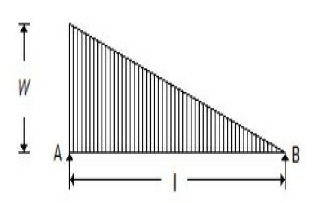
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
The total elongation produced in a bar of uniform section hanging vertically downwards due to its own weight is equal to that produced by a weight
Of same magnitude as that of bar and applied at the lower end
Half the weight of bar applied at lower end
Half of the square of weight of bar applied at lower end
One fourth of weight of bar applied at lower end
A process, in which the working substance neither receives nor gives out heat to its surroundings during its expansion or compression, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
The stress at which extension of the material takes place more quickly as compared to the increase in load is called
Elastic point of the material
Plastic point of the material
Breaking point of the material
Yielding point of the material
Which is the false statement about true stress strain method?
It does not exist
It is more sensitive to changes in both metallurgical and mechanical conditions
It gives a more accurate picture of the ductility
It can be correlated with stress strain values in other tests like torsion, impact, combined stress tests etc.
The ratio of specific heat at constant pressure (cp) and specific heat at constant volume (cv) is always __________ one.
Equal to
Less than
Greater than
None of these
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
The unit of energy is S. I. units is
Joule (J)
Joule metre (Jm)
Watt (W)
Joule/metre (J/m)
In a steady flow process, the ratio of
Heat transfer is constant
Work transfer is constant
Mass flow at inlet and outlet is same
All of these
The efficiency of Joule cycle is
Greater than Carnot cycle
Less than Carnot cycle
Equal to Carnot cycle
None of these
The value of Poisson's ratio for steel is between
0.01 to 0.1
0.23 to 0.27
0.25 to 0.33
0.4 to 0.6
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
The amount of heat required to raise the temperature of the unit mass of gas through one degree at constant volume, is called
Specific heat at constant volume
Specific heat at constant pressure
Kilo Joule
None of these
A body is subjected to a direct tensile stress of 300 MPa in one plane accompanied by a simple shear stress of 200 MPa. The maximum shear stress will be
-100 MPa
250 MPa
300 MPa
400 MPa
The fuel mostly used in blast furnace for extracting pig iron from iron ores is
Hard coke
Soft coke
Pulverised coal
Bituminous coal
The entropy of water at 0°C is assumed to be
1
0
-1
10
If Kh is the torque resisting capacity of a hollow shaft and Ks is that of a solid shaft, of the same material, length and weight. Then,
Kh > Ks
Kh < Ks
Kh = Ks
None of these
The bending moment of a cantilever beam of length l and carrying a uniformly distributed load of w per unit length is __________ at the fixed end.
wl/4
wl/2
wl
wl²/2
The gas constant (R) is equal to the __________ of two specific heats.
Sum
Difference
Product
Ratio
During which of the following process does heat rejection takes place in Carnot cycle?
Isothermal expansion
Isentropic expansion
Isothermal compression
Isentropic compression
