The heat capacities for the ideal gas state depend upon the
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
Correct Answer :
B. Temperature
Related Questions
Pick out the wrong statement.
Enthalpies of all elements in their standard states are assumed to be zero
Combustion reactions are never endothermic in nature
Heat of reaction at constant volume is equal to the change in internal energy
Clausius-Clapeyron equation is not applicable to melting process
The entropy change in a reversible isothermal process, when an ideal gas expands to four times its initial volume is
R loge 4
R log10 4
Cv log10 4
Cv loge 4
(1/V) (∂V/∂T)P is the mathematical expression
Joule-Thomson co-efficient
Specific heat at constant pressure (Cp)
co-efficient of thermal expansion
Specific heat at constant volume (CV)
A/an __________ system is exemplified by a vessel containing a volatile liquid in contact with its vapor.
Isolated
Closed
Open
None of these
For organic compounds, group contribution method can be used for the estimation of
Critical properties
Specific gravity
Specific volume
Thermal conductivity
(∂H/∂T)P is the mathematical expression for
CV
Entropy change
Gibbs free energy
None of these
At __________ point, all the three phases (i.e. solid, liquid and gas) co-exist.
Eutectic
Triple
Plait
Critical
Specific/molar Gibbs free energy for a pure substance does not change during
Sublimation
Vaporisation
Melting
Either (A), (B) or (C)
__________ explains the equilibrium constant for any chemical reaction.
Henry's law
Law of mass action
Hess's law
None of these
Normal temperature and pressure (N.T.P.) corresponds to
0°C and 760 mm Hg
15°C and 760 mm Hg
20°C and 760 mm Hg
0°C and 1 kgf/cm2
Pick out the extensive property out of the following.
Surface tension
Free energy
Specific heat
Refractive index
Consider the process A & B shown in the figure given below: In this case, it is possible that
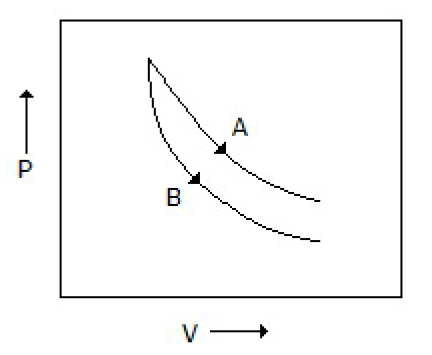
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
Melting of wax is accompanied with __________ in entropy.
Increase
Decrease
No change
None of these
Which of the following is not a unit of the equilibrium constant Kp? (where, Δx = number of moles of products number of moles of reactants)
(atm)Δx, when Δx is negative
(atm)Δx, when Δx is positive
Dimensionless, when Δx = 0
(atm)Δx2, when Δx > 0
Cp of a gas at its critical temperature and pressure
Becomes zero
Becomes infinity
Equals 1 kcal/kmol °K
Equals 0.24 kcal/kmol °K
First law of thermodynamics is mathematically stated as
dQ = dE + dW
dQ = dE - dW
dE = dQ + dW
dW = dQ + dE
__________ law of thermodynamics ascertains the direction of a particular spontaneous process.
Zeroth
First
Second
Third
Maximum work that could be secured by expanding the gas over a given pressure range is the __________ work.
Isothermal
Adiabatic
Isentropic
None of these
Which is an example of closed system?
Air compressor
Liquid cooling system of an automobile
Boiler
None of these
1st law of thermodynamics is nothing but the law of conservation of
Momentum
Mass
Energy
None of these
For a constant volume process __________ by the system is used only to increase the internal energy.
Heat absorbed
Work done
Both (A) & (B)
Neither (A) nor (B)
The free energy change for a chemical reaction is given by (where, K = equilibrium constant)
RT ln K
-RT ln K
-R ln K
T ln K
Which of the following units is not present in both the vapor compression refrigeration system and absorption refrigeration system?
Expansion valve
Condenser
Refrigerator
Compressor
The melting point of paraffin wax (which contracts on solidification) __________ with pressure rise.
Increases
Decreases
Remains unchanged
Decreases linearly
Chemical potential (an intensive property) of a substance is a force that drives the chemical system to equilibrium and is equal to its partial molar properties. The ratio of chemical potential to free energy of a pure substance at constant temperature and pressure is
0
1
∞
None of these
Gibbs free energy of a pure fluid approaches __________ as the pressure tends to zero at constant temperature.
Infinity
Minus infinity
Zero
None of these
The enthalpy change when ammonia gas is dissolved in water is called the heat of
Solution
Formation
Dilution
Combustion
Filling of gas from a high pressure cylinder into small bottles is an example of a/an __________ process.
Equilibrium
Adiabatic
Steady
Unsteady
The chemical potential of any constituent of an ideal solution depends on the __________ of the solution.
Temperature
Pressure
Composition
All (A), (B) and (C)
An irreversible process
Is the analog of linear frictionless motion in machines
Is an idealised visualisation of behaviour of a system
Yields the maximum amount of work
Yields an amount of work less than that of a reversible process
