The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
Correct Answer :
C. Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Related Questions
A cycle consisting of two isothermal and two isentropic processes, is known as
Carnot cycle
Stirling cycle
Ericsson cycle
Joule cycle
If Kh is the torque resisting capacity of a hollow shaft and Ks is that of a solid shaft, of the same material, length and weight. Then,
Kh > Ks
Kh < Ks
Kh = Ks
None of these
The materials which exhibit the same elastic properties in all directions are called
Homogeneous
Inelastic
Isotropic
Isentropic
Petrol is distilled at
65° to 220°C
220° to 345°C
345° to 470°C
470° to 550°C
The efficiency of Diesel cycle increases with
Decrease in cut-off
Increase in cut-off
Constant cut-off
None of these
Relation between cp and cv is given by (where cp = Specific heat at constant pressure, cv = Specific heat at constant volume, γ = cp/cv, known as adiabatic index, and R = Gas constant)
cv/ cp =R
cp - cv = R
cv = R/ γ-1
Both (B) and (C)
The universal gas constant (or molar constant) of a gas is the product of
Molecular mass of the gas and the gas constant
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the specific heat at constant pressure
Molecular mass of the gas and the specific heat at constant volume
One kg of carbon monoxide requires 4/7 kg of oxygen and produces
11/3 kg of carbon dioxide gas
7/3 kg of carbon monoxide gas
11/7 kg of carbon dioxide gas
8/3 kg of carbon monoxide gas
The shear force at the centre of a simply supported beam with a gradually varying load from zero at both ends to w per metre at the centre, is
Zero
wl/4
wl/2
wl²/2
The value of gas constant (R) in S. I. units is
0.287 J/kgK
2.87 J/kgK
28.7 J/kgK
287 J/kgK
The weakest section of a diamond riveting is the section which passes through
The first row
The second row
The central row
One rivet hole of the end row
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
In the tensile test, the phenomenon of slow extension of the material, i. e. stress increasing with the time at a constant load is called
Creeping
Yielding
Breaking
Plasticity
The maximum bending moment for the beam shown in the below figure, is
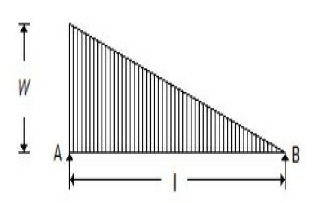
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
For the beam shown in the below figure, the shear force diagram between A and B is
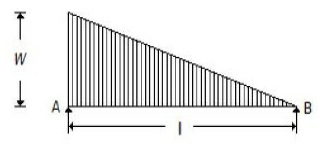
A horizontal line
A vertical line
An inclined line
A parabolic curve
The amount of heat generated per kg of fuel is known as
Calorific value
Heat energy
Lower calorific value
Higher calorific value
A continuous beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported on more than two supports
Extending beyond the supports
Mond gas is obtained by
Partial combustion of coal, coke, anthracite coal or charcoal in a mixed air steam blast
Carbonisation of bituminous coal
Passing steam over incandescent coke
Passing air and a large amount of steam over waste coal at about 650°C
The assumption made in Euler's column theory is that
The failure of column occurs due to buckling alone
The length of column is very large as compared to its cross-sectional dimensions
The column material obeys Hooke's law
All of the above
The molecular mass expressed in gram (i.e. 1 g - mole) of all gases, at N. T. P., occupies a volume of
0.224 litres
2.24 litres
22.4 litres
224 litres
If the slenderness ratio for a column is 100, then it is said to be a _________ column.
Long
Medium
Short
None of these
The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
The property of a working substance which increases or decreases as the heat is supplied or removed in a reversible manner, is known as
Enthalpy
Internal energy
Entropy
External energy
The absolute zero pressure will be
When molecular momentum of the system becomes zero
At sea level
At the temperature of - 273 K
At the centre of the earth
The compression ratio is the ratio of
Swept volume to total volume
Total volume to swept volume
Swept volume to clearance volume
Total volume to clearance volume
The efficiency of Joule cycle is
Greater than Carnot cycle
Less than Carnot cycle
Equal to Carnot cycle
None of these
A process of heating crude oil to a high temperature under a very high pressure to increase the yield of lighter distillates, is known as
Cracking
Carbonisation
Fractional distillation
Full distillation
An isothermal process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The ideal efficiency of a Brayton cycle with regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
The air standard efficiency of an Otto cycle is given by (where r = Compression ratio, and γ = Ratio of specific heats)
1 - rγ - 1
1 + rγ - 1
1 - (1/ rγ - 1)
1 + (1/ rγ - 1)
