The specific heat at constant volume is
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant pressure
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
The amount of heat required to raise the temperature of 1 kg of water through one degree
Any one of the above
Correct Answer :
B. The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
Related Questions
When gas is heated at constant pressure, the heat supplied is utilised in
Increasing the internal energy of gas
Doing some external work
Increasing the internal energy of gas and also for doing some external work
None of the above
Brayton cycle consists' of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
A masonry dam may fail due to
Tension in the masonry of the dam and its base
Overturning of the dam
Crushing of masonry at the base of the dam
Any one of the above
Volumetric strain for a rectangular specimen of length l, breadth b and thickness t subjected to a pull of P is given by
e (1 - 2m)
e (1 - 2/m)
e (m - 2)
e (2/m - 1)
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
For which material the Poisson's ratio is more than unity
Steel
Copper
Aluminium
None of the above
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
The pull required to tear off the plate per pitch length is (where p = Pitch of rivets, t = Thickness of plates, and σt, τ and σc = Permissible tensile, shearing and crushing stresses respectively)
(p - 2d) t × σc
(p - d) t × τ
(p - d) t × σt
(2p - d) t × σt
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
A key is subjected to side pressure as well at shearing forces. These pressures are called
Bearing stresses
Fatigue stresses
Crushing stresses
Resultant stresses
In open cycle gas turbine plants
The indirect heat exchanger and cooler is avoided
Direct combustion system is used
A condenser is used
All of the above
In S. I. units, the value of the universal gas constant is
8.314 J/kg mole-K
83.14 J/kgmole-K
831.4 J/kgmole-K
8314 J/kgmole-K
The limit of eccentricity for no tensile conditions for a column of circular section of diameter (D) is
d/4
d/8
d/12
d/16
The fuel mostly used in cement industry and in metallurgical processes is
Wood charcoal
Bituminous coke
Pulverised coal
Coke
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
Modular ratio of two materials is the ratio of
Strains
Stress and strain
Shear stress and shear strain
Moduli and elasticity
When the gas is cooled at constant pressure,
Its temperature increases but volume decreases
Its volume increases but temperature decreases
Both temperature and volume increases
Both temperature and volume decreases
A process of heating crude oil to a high temperature under a very high pressure to increase the yield of lighter distillates, is known as
Cracking
Carbonisation
Fractional distillation
Full distillation
A mixture of gas expands from 0.03 m3 to 0.06 m3 at a constant pressure of 1 MPa and absorbs 84 kJ of heat during the process. The change in internal energy of the mixture is
30 kJ
54 kJ
84 kJ
114 kJ
In the below figure, the plastic range occurs
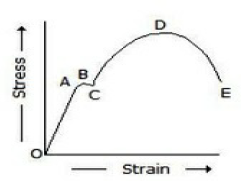
Before point A
Beyond point A
Between points A and D
Between points D and E
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
Resilience is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of the specimen maximum strain
Energy which can be stored in a body
None of the above
The state of a substance whose evaporation from its liquid state is complete, is known as
Vapour
Perfect gas
Air
Steam
Which of the following is correct?
Gauge pressure = Absolute pressure + Atmospheric pressure
Absolute pressure = Gauge pressure + Atmospheric pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
The ratio of elongation in a prismatic bar due to its own weight (W) as compared to another similar bar carrying an additional weight (W) will be
1 : 2
1 : 3
1 : 4
1 : 2.5
An open system is one in which
Heat and work crosses the boundary of the system, but the mass of the working substance does not crosses the boundary of the system
Mass of the working substance crosses the boundary of the system but the heat and work does not crosses the boundary of the system
Both the heat and work as well as mass of the working substance crosses the boundary of the system
Neither the heat and work nor the mass of the working substance crosses the boundary of the system
When a rectangular beam is loaded transversely, the maximum compressive stress is developed on the
Top layer
Bottom layer
Neutral axis
Every cross-section
Two shafts 'A' and 'B' transmit the same power. The speed of shaft 'A' is 250 r.p.m. and that of shaft 'B' is 300 r.p.m.
The shaft 'B' has the greater diameter
The shaft 'A' has the greater diameter
Both are of same diameter
None of these
Which of the following cycles is not a reversible cycle?
Carnot
Ericsson
Stirling
None of the above
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
