The two elements required to form substitutional solid solution should not have
Same crystalline structure
Same valency
Widely differing electronegativity
Same atomic sizes
Correct Answer :
C. Widely differing electronegativity
Related Questions
More than 95% of __________ is present in corundum.
SiO2
Al2O3
CaSiO2
MgO
For infinite parallel planes having emissivities ε1 & ε2, the interchange factor for radiation from surface 1 to surface 2 is given by
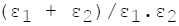
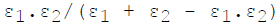
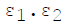
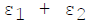
Heat release during phase change is observed in case of a/an
Boiler
Condenser
Evaporator
All (A), (B) & (C)
Pick out the correct statement.
Materials exhibiting high elasticity obey Hooke's law
The elastic behaviour of rubber under compression is the same as its behaviour under tension
The damping capacity of a material is due to its plastic deformation
The stress required to cause plastic flow in polycrystalline material is higher as compared to monocrystalline materials due to the presence of grains of different orientations
The most commonly used moderator in nuclear power plants is
Graphite
Light water
Heavy water
Beryllium
Upto the critical radius of insulation, added insulation, will
Increase the heat loss
Decrease heat loss and will have no effect beyond that
Decrease heat flux
Reduce convection heat loss compared to conduction heat less
What is the value of entropy at 273°K?
0
1
∞
None of these
Pearlite comprises of
87% cementite & 13% Fe
87% ferrite & 13% cementite
93.33% ferrite & 6.67% C
87% ferrite & 13% C
Cast iron contains __________ percent carbon.
2.5
3.75
4.75
5.25
Maximum change in the hardness of martensite occurs in the carbon content range of __________ percent in steel.
0.01 to 0.10
0.2 to 0.4
0.5 to 0.7
0.8 to 1.0
Primary designation of steel is based on its
Hardness & tensile strength
Carbon content
Iron content
Alloying elements content
Hot & cold working of material causes its __________ deformation.
Visco-elastic
Isotropic
Elastic
Plastic
__________ of air does not increase with increase in temperature.
Density
Thermal diffusivity
Viscosity
Thermal conductivity
Dryness fraction of dry steam is
0
∞
1
0.5
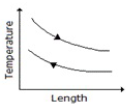
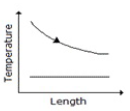
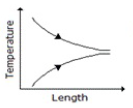
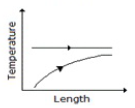
Temperature profile along the length of a gasgas counter flow heat exchanger is correctly represented by
Sacrificial anode method is used in the protection of pipelines which are buried underground. Sacrificial anode
Occurs higher in the electro-chemical series of metals
Is exemplified by magnesium plate
Is much cheaper than the cathodic base material to be guarded against corrosion
All (A), (B) & (C)
Hot extrusion process is not used for making
Cast iron pipe for domestic water supply
Aluminium curtain rods
Stainless steel tubes used in furnitures
Any of these
Which of the following is not a ferromagnetic material?
Nickel
Cobalt
Aluminium
Iron
Identify the correct statement with reference to the extractive metallurgy of aluminium.
The electrolyte consists of molten Na3AlF6 with approximately 1 to 8% Al2O3
Approximately 80% of the aluminium deposited on the cathode comes from cryolite
Sodium is deposited along with aluminium, but is immediately vaporised
Anode effect sets in when the cryolite concentration goes below 40%.
Maximum permissible air velocity in pipelines is about __________ metre/second.
5
10
20
40
The noble metals
Are exemplified by Na, K and Li
Do not resist corrosion very strongly
Are the lowermost in the electro-chemical series of metals
None of these
Brittleness induced due to the presence of sulphur in steel can be reduced by adding
Manganese
Magnesium
Vanadium
Copper
The best lubricants for a machine working at high temperature & load is
Grease
Solid lubricant
Animal oils
Mineral oils
Window panes of aeroplanes are normally made of
Perspex (PMMA)
Teflon (PTFE)
Bakelite (phenol formaldehyde)
Polystyrene
A furnace is made of a refractory brick wall of thickness 0.5 metre and thermal conductivity 0.7 W/m.°K For the same temperature drop and heat loss, this refractory wall can be replaced by a layer of diatomaceous earth of thermal conductivity 0.14 W/m.K and thickness __________ metre.
0.01
0.1
0.25
0.5
Copper deposits are found in India at the following location:
Kudremukh
Kolar
Khetri
Ramagundam
Minimum safe distance between two liquid fuel storage tanks is equal to (where, H = height of the tank)
H
H/2
H/4
H/6
The elastic strain in copper is due to the
Motion of dislocations
Stretching of atomic bonds
Breakage of atomic bonds
None of the above
Lead is poured into the joint between two- __________ pipes.
Mild steel
Concrete
Cast iron
Asbestos cement
In a drilling process, the metal is removed by both shearing & extrusion. General purpose drills are made of
Stainless steel
Mild steel
High speed steel
High carbon steel
