What is the degree of freedom for two miscible (non-reacting) substances in vapor-liquid equilibrium forming an azeotrope?
0
1
2
3
Correct Answer :
C. 2
Related Questions
With increase in compression ratio, the efficiency of the otto engine
Increases
Decreases
Remain constant
Increases linearly
Change of heat content when one mole of compound is burnt in oxygen at constant pressure is called the
Calorific value
Heat of reaction
Heat of combustion
Heat of formation
COP of a refrigerator drawing 1 kW of power per ton of refrigeration is about
0.5
3.5
4.5
8.5
In polytropic process (PVn = constant), if n = 1; it means a/an __________ process.
Adiabatic
Reversible
Isothermal
None of these
The unit of equilibrium constant of a chemical reaction is the same as that of
Molar concentration
Temperature
Internal energy
None of these
The difference between isothermal compressibility and adiabatic compressibility for an ideal gas is
0
+ve
-ve
∞
Trouton's ratio of __________ liquids is calculated using Kistyakowsky equation.
Polar
Non-polar
Both (A) & (B)
Neither (A) nor (B)
A thermodynamic system is taken from state A to B along ACB and is brought back to A along BDA as shown below in the P-V diagram. The net work done during the complete cycle is given by the area covered by

P1ACBP2P1
ACBB1A1A
ACBDA
ADBB1A1A
Out of the following refrigeration cycles, which one has maximum COP?
Air cycle
Carnot cycle
Ordinary vapor compression cycle
Vapor compression with a reversible expansion engine
One ton of refrigeration capacity is equivalent to the heat removal rate of
50 kcal/hr
200 BTU/hr
200 BTU/minute
200 BTU/day
Air enters an adiabatic compressor at 300K. The exit temperature for a compression ratio of 3, assuming air to be an ideal gas (Y = Cp/Cv = 7/5) and the process to be reversible, is
300 × (32/7)
300 × (33/5)
300 × (333/7)
300 × (35/7)
The ammonia synthesis reaction represented by N2 + 3H2 2NH3; ΔH = - 22.4 kcal, is
Endothermic
Exothermic
Isothermal
Adiabatic
The necessary condition for phase equilibrium in a multiphase system of N components is that the
Chemical potentials of a given component should be equal in all phases
Chemical potentials of all components should be same in a particular phase
Sum of the chemical potentials of any given component in all the phases should be the same
None of these
Refrigerants commonly used for domestic refrigerators are
Ethyl chloride or methyl chloride
Freon-12
Propane
NH3 or CO2
The internal energy of a gas obeying P (V - b) RT (where, b is a positive constant and has a constant Cv), depends upon its
Pressure
Volume
Temperature
All (A), (B) & (C)
Consider the process A & B shown in the figure given below: In this case, it is possible that
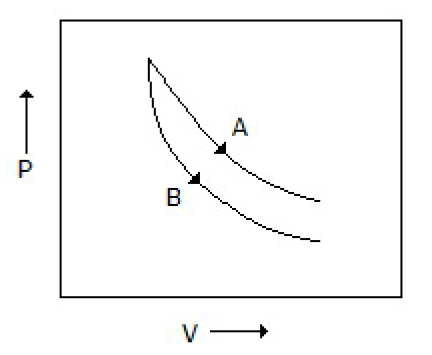
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
If different processes are used to bring about the same chemical reaction, the enthalpy change is same for all of them. This is __________ law.
Hess's
Kirchoff's
Lavoisier and Laplace
None of these
Out of the following refrigeration cycles, which one has the minimum COP (Co-efficient of performance)?
Air cycle
Carnot cycle
Ordinary vapour compression cycle
Vapour compression with a reversible expansion engine
Pick out the correct statement:
In an isothermal system, irreversible work is more than reversible work
Under reversible conditions, the adiabatic work is less than isothermal work
Heat, work, enthalpy and entropy are all 'state functions'
Matter and energy cannot be exchanged with the surroundings in a closed system
For a constant pressure reversible process, the enthalpy change (ΔH) of the system is
Cv.dT
Cp.dT
∫ Cp.dT
∫ Cv.dT
To obtain integrated form of Clausius-Clapeyron equation, ln (P2/P1) = (ΔHV/R) (1/T1 - 1/T2) from the exact Clapeyron equation, it is assumed that the
Volume of the liquid phase is negligible compared to that of vapour phase
Vapour phase behaves as an ideal gas
Heat of vaporisation is independent of temperature
All (A), (B) & (C)
Measurement of thermodynamic property of temperature is facilitated by __________ law of thermodynamics.
1st
Zeroth
3rd
None of these
The specific heat of saturated water vapour at 100°C is
∞
-ve
0
+ve
Extensive properties of a thermodynamic system depend upon the __________ of the system.
Specific volume
Temperature
Mass
Pressure
Pick out the correct statement.
Compression ratio of an Otto engine is comparatively higher than a diesel engine
Efficiency of an Otto engine is higher than that of a diesel engine for the same compression ratio
Otto engine efficiency decreases with the rise in compression ratio, due to decrease in work produced per quantity of heat
Diesel engine normally operates at lower compression ratio than an Otto engine for an equal output of work
Charles' law for gases states that
V/T = Constant
V ∝ 1/T
V ∝ 1/P
PV/T = Constant
Gibbs-Helmholtz equation is
ΔF = ΔH + T [∂(ΔF)/∂T]P
ΔF = ΔH - TΔT
d(E - TS) T, V < 0
dP/dT = ΔHvap/T.ΔVvap
When a system is in equilibrium for all possible processes, the differential or finite change of entropy is
< 0
> 0
= 0
None of these
Which of the following is not an intensive property?
Chemical potential
Surface tension
Heat capacity
None of these
The internal energy of an ideal gas is a function of its __________ only.
Molecular size
Volume
Pressure
Temperature
