When a gas in a vessel expands, its internal energy decreases. The process involved is
Reversible
Irreversible
Isothermal
Adiabatic
Correct Answer :
A. Reversible
Related Questions
Entropy change for an irreversible process taking system and surrounding together is
0
> 0
< 0
None of these
Which of the following will increase the volume of a real gas by four times?
Doubling the absolute temperature as well as pressure of the gas
Reducing pressure to one fourth at constant temperature
Reducing temperature to one fourth at constant pressure
Reducing the temperature to half and doubling the pressure
Trouton's ratio is given by (where λb, = molal heat of vaporisation of a substance at its normal boiling point, kcal/kmol Tb = normal boiling point, °K)
λb/Tb
Tb/λb
√(λb/Tb)
√(Tb/λb)
As pressure approaches zero, the ratio of fugacity to pressure (f/P) for a gas approaches
Zero
Unity
Infinity
An indeterminate value
COP of a refrigerator drawing 1 kW of power per ton of refrigeration is about
0.5
3.5
4.5
8.5
The molar excess Gibbs free energy, gE, for a binary liquid mixture at T and P is given by, (gE/RT) = A . x1. x2, where A is a constant. The corresponding equation for ln y1, where y1 is the activity co-efficient of component 1, is
A . x22
Ax1
Ax2
Ax12
The internal energy of an incompressible fluid depends upon its
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
The point at which all the three (solid, liquid and gas) phases co-exist, is known as the __________ point.
Freezing
Triple
Boiling
Boyle
Gases are cooled in Joule-Thomson expansion, when it is __________ inversion temperature.
Below
At
Above
Either 'b' or 'c'
At constant temperature and pressure, for one mole of a pure substance, the ratio of the free energy to the chemical potential is
Zero
One
Infinity
Negative
Van Laar equation deals with the activity coefficients in
Binary solutions
Ternary solutions
Azeotropic mixture only
None of these
(∂H/∂T)P is the mathematical expression for
CV
Entropy change
Gibbs free energy
None of these
To obtain integrated form of Clausius-Clapeyron equation, ln (P2/P1) = (ΔHV/R) (1/T1 - 1/T2) from the exact Clapeyron equation, it is assumed that the
Volume of the liquid phase is negligible compared to that of vapour phase
Vapour phase behaves as an ideal gas
Heat of vaporisation is independent of temperature
All (A), (B) & (C)
Which of the following is affected by the temperature?
Fugacity
Activity co-efficient
Free energy
All (A), (B) & (C)
Kopp's rule is used to calculate the heat capacity of
Solids
Liquids
Gases
All (A), (B) & (C)
Joule-Thomson experiment is
Isobaric
Adiabatic
Isenthalpic
Both (B) & (C)
Pick out the wrong statement.
Activity co-efficient is dimensionless.
In case of an ideal gas, the fugacity is equal to its pressure.
In a mixture of ideal gases, the fugacity of a component is equal to the partial pressure of the component.
The fugacity co-efficient is zero for an ideal gas
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
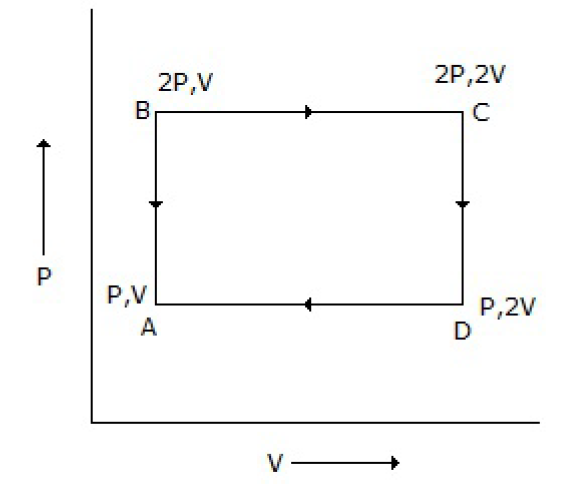
PV
2PV
PV/2
0
Efficiency of a Carnot engine working between temperatures T1 and T2 (T1 < T) is
(T2 - T1)/T2
(T2 - T1)/T1
(T1 - T2)/T2
(T1 - T2)/T1
In an ideal solution, the activity of a component equals its
Mole fraction
Fugacity at the same temperature and pressure
Partial pressure
None of these
Solubility of a substance which dissolves with an increase in volume and liberation of heat will be favoured by the
Low pressure and high temperature
Low pressure and low temperature
High pressure and low temperature
High pressure and high temperature
Generation of heat by friction is an example of a/an __________ change.
Isothermal
Irreversible
Adiabatic
Reversible
An irreversible process
Is the analog of linear frictionless motion in machines
Is an idealised visualisation of behaviour of a system
Yields the maximum amount of work
Yields an amount of work less than that of a reversible process
The most important application of distribution law is in
Evaporation
Liquid extraction
Drying
Distillation
4 kg moles of an ideal gas expands in vacuum spontaneously. The work done is
4 J
∞
0
8 J
All gases during throttling process at atmospheric temperature and pressure show a cooling effect except
CO2
H2
O2
N2
The internal energy of an ideal gas does not change in a reversible __________ process.
Isothermal
Adiabatic
Isobaric
Isometric
Extensive properties of a thermodynamic system depend upon the __________ of the system.
Specific volume
Temperature
Mass
Pressure
Pick out the wrong statement:
The expansion of a gas in vacuum is an irreversible process
An isometric process is a constant pressure process
Entropy change for a reversible adiabatic process is zero
Free energy change for a spontaneous process is negative
The melting point of paraffin wax (which contracts on solidification) __________ with pressure rise.
Increases
Decreases
Remains unchanged
Decreases linearly
