When liquid and vapour phases of one component system are in equilibrium (at a given temperature and pressure), the molar free energy is
More in vapour phase
More in liquid phase
Same in both the phases
Replaced by chemical potential which is more in vapour phase
Correct Answer :
C. Same in both the phases
Related Questions
Choose the condition that must be specified in order to liquify CO2 (triple point for CO2 is - 57°C and 5.2 atm).
Pressure must be kept below 5.2 atm
Temperature must be kept above - 57°C
Pressure must be kept below 5.2 atm. and temperature must be kept above 57°C
Pressure and temperature must be kept below 5.2 atm. and - 57°C respectively
__________ decreases during adiabatic throttling of a perfect gas.
Entropy
Temperature
Enthalpy
Pressure
In a P-V diagram (for an ideal gas), an isothermal curve will coincide within adiabatic curve (through a point), when
Cp < Cv
Cp = Cv
Cp > Cv
C ≥ Cv
__________ Equation predicts the activity coefficient from experimental data.
Lewis-Randall
Margules
Van Laar
Both (B) & (C)
For a given substance at a specified temperature, activity is __________ to fugacity.
Directly proportional
Inversely proportional
Equal
None of these
Chemical engineering thermodynamics is concerned with the __________ in/of chemical processes.
Reaction mechanism
Calculation of rates
Energy transformation from one form to another
None of these
Compound having large heat of formation is
More stable
Less stable
Not at all stable (like nascent O2)
Either more or less stable; depends on the compound
The third law of thermodynamics states that the
Heat capacity of a crystalline solid is zero at absolute zero temperature
Heat transfer from low temperature to high temperature source is not possible without external work
Gases having same reduced properties behaves similarly
None of these
Specific volume of an ideal gas is
Equal to its density
The reciprocal of its density
Proportional to pressure
None of these
Boiling of liquid is accompanied with increase in the
Vapor pressure
Specific Gibbs free energy
Specific entropy
All (A), (B) and (C)
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
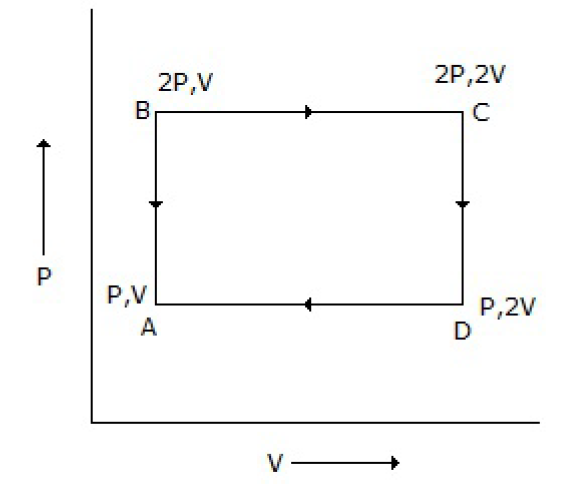
PV
2PV
PV/2
0
There is a change in __________ during the phase transition.
Volume
Pressure
Temperature
All a, b & c
During the phase transition, __________ changes.
Pressure
Volume
Temperature
All (A), (B) and (C)
Minimum number of phases that exists in a system is 1. Number of chemical species in a colloidal system is
1
2
3
4
At a given temperature, the volume of a gas dissolved in a solvent __________ with increase in pressure.
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the gas
If an ideal solution is formed by mixing two pure liquids in any proportion, then the __________ of mixing is zero
Enthalpy
Volume
Both 'a' & 'b'
Neither 'a' nor 'b'
For the reversible exothermic reaction, N2 + 3H2 2NH3, increase of pressure would
Shift the equilibrium towards right
Give higher yield of NH3
Both (B) and (C)
Neither (A) nor (B)
A thermodynamic system is taken from state A to B along ACB and is brought back to A along BDA as shown below in the P-V diagram. The net work done during the complete cycle is given by the area covered by

P1ACBP2P1
ACBB1A1A
ACBDA
ADBB1A1A
Isobaric process means a constant process.
Temperature
Pressure
Volume
Entropy
Third law of thermodynamics is helpful in
Prediction of the extent of a chemical reaction
Calculating absolute entropies of substances at different temperature
Evaluating entropy changes of chemical reaction
Both (B) and (C)
In a homogeneous solution, the fugacity of a component depends upon the
Pressure
Composition
Temperature
All (A), (B) and (C)
Which of the following equations is obtained on combining 1st and 2nd law of thermodynamics, for a system of constant mass?
dE = Tds - PdV
dQ = CvdT + PdV
dQ = CpdT + Vdp
Tds = dE - PdV
For a thermodynamic system containing 'x' chemical species, the maximum number of phases that can co-exist at equilibrium is
x
x + 1
x + 2
x + 3
At the absolute zero temperature, the entropy of every perfectly crystalline substance becomes zero. This follows from the
Third law of thermodynamics
Second law of thermodynamics
Nernst heat theorem
Maxwell's relations
For a multi-component system, the term chemical potential is equivalent to the
Molal concentration difference
Molar free energy
Partial molar free energy
Molar free energy change
For an exothermic reaction
Only enthalpy change (ΔH) is negative
Only internal energy change (ΔE) is negative
Both ΔH and ΔE are negative
Enthalpy change is zero
Internal energy is equal to the heat absorbed in case of a/an __________ process.
Constant volume
Polytropic
Adiabatic
Constant pressure
Efficiency of a Carnot engine working between temperatures T1 and T2 (T1 < T) is
(T2 - T1)/T2
(T2 - T1)/T1
(T1 - T2)/T2
(T1 - T2)/T1
The change in __________ is equal to the reversible work for compression in steady state flow process under isothermal condition.
Internal energy
Enthalpy
Gibbs free energy
Helmholtz free energy
What is the degree of freedom for two miscible (non-reacting) substances in vapor-liquid equilibrium forming an azeotrope?
0
1
2
3
