When the gas is cooled at constant pressure,
Its temperature increases but volume decreases
Its volume increases but temperature decreases
Both temperature and volume increases
Both temperature and volume decreases
Correct Answer :
D. Both temperature and volume decreases
Related Questions
A mixture of gas expands from 0.03 m3 to 0.06 m3 at a constant pressure of 1 MPa and absorbs 84 kJ of heat during the process. The change in internal energy of the mixture is
30 kJ
54 kJ
84 kJ
114 kJ
The stress induced in a body, when suddenly loaded, is ________ the stress induced when the same load is applied gradually.
Equal to
One-half
Twice
Four times
Which of the following is an irreversible cycle?
Carnot
Stirling
Ericsson
None of the above
The unit of energy is S. I. units is
Joule (J)
Joule metre (Jm)
Watt (W)
Joule/metre (J/m)
Which of the following gas has a minimum molecular mass?
Oxygen
Nitrogen
Hydrogen
Methane
An open cycle gas turbine works on
Carnot cycle
Otto cycle
Joule's cycle
Stirling cycle
A circular shaft fixed at, A has diameter D for half of its length and diameter D/2 over the other half, as shown in the below figure. If the rotation of B relative to A is 0.1 radian, the rotation of C relative to B will be
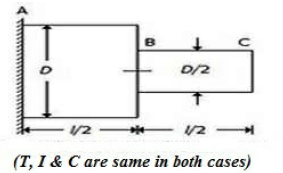
0.4 radian
0.8 radian
1.6 radian
3.2 radian
Reversed Joule cycle is known as
Carnot cycle
Bell-Coleman cycle
Rankine cycle
Stirling cycle
The smallest quantity of a substance, which can exist by itself in a chemically recognizable form is known as
Element
Compound
Atom
Molecule
According to Euler's column theory, the crippling load for a column of length (l) with one end fixed and the other end free is __________ the crippling load for a similar column hinged at both the ends.
Equal to
Less than
More than
None of these
The specific heat at constant volume is
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant pressure
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
The amount of heat required to raise the temperature of 1 kg of water through one degree
Any one of the above
Otto cycle is also known as
Constant pressure cycle
Constant volume cycle
Constant temperature cycle
Constant temperature and pressure cycle
Which of the following has the highest calorific value?
Peat
Lignite
Bituminous coal
Anthracite coal
Carnot cycle has maximum efficiency for
Petrol engine
Diesel engine
Reversible engine
Irreversible engine
When shear force at a point is zero, then bending moment is _________ at that point.
Zero
Minimum
Maximum
Infinity
The deformation per unit length is called
Tensile stress
Compressive stress
Shear stress
Strain
The efficiency of a Carnot engine depends on
Working substance
Design of engine
Size of engine
Temperatures of source and sink
The oxygen atom is ________ times heavier than the hydrogen atom.
2
8
16
32
A cube subjected to three mutually perpendicular stress of equal intensity p expenses a volumetric strain
3p/E × (2/m - 1)
3p/E × (2 - m)
3p/E × (1 - 2/m)
E/3p × (2/m - 1)
The bending moment at a point on a beam is the algebraic ________ of all the moments on either side of the point.
Sum
Difference
Multiplication
None of the above
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
The total energy of a molecule is shared equally by the various degrees of freedom possessed by it. This law is known as
Law of equipartition of energy
Law of conservation of energy
Law of degradation of energy
None of these
Strain energy is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of a specimen
Maximum strain energy which can be stored in a body
Proof resilience per unit volume of a material
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
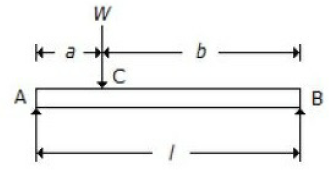
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
In a belt drive, the pulley diameter is doubled, the belt tension and pulley width remaining same. The changes required in key will be
Increase key length
Increase key depth
Increase key width
Double all the dimensions
If a part is constrained to move and heated, it will develop
Principal stress
Tensile stress
Compressive stress
Shear stress
The specific heat of water is
1.817
2512
4.187
None of these
A bar of copper and steel form a composite system, which is heated to a temperature of 40°C. The stress induced in the copper bar will be
Tensile
Compressive
Shear
Zero
Otto cycle efficiency is higher than Diesel cycle efficiency for the same compression ratio and heat input because in Otto cycle
Combustion is at constant volume
Expansion and compression are isentropic
Maximum temperature is higher
Heat rejection is lower
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
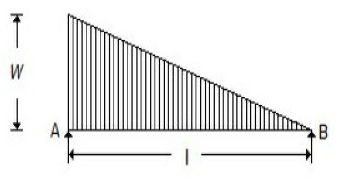
wl/6
wl/3
wl
2wl/3
