When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
Correct Answer :
D. Both (B) and (C)
Related Questions
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
The expansion ratio (r) is the ratio of (where v1 = Volume at the beginning of expansion, and v2 = Volume at the end of expansion)
v1/v2
v2/v1
(v1 + v2)/v1
(v1 + v2)/v2
A cube subjected to three mutually perpendicular stress of equal intensity p expenses a volumetric strain
3p/E × (2/m - 1)
3p/E × (2 - m)
3p/E × (1 - 2/m)
E/3p × (2/m - 1)
Formula based on IS codes is based on
Straight line formula
Eulers formula
Rankines formula
Secant formula
The value of 1 mm of Hg is equal to
1.333 N/m2
13.33 N/m2
133.3 N/m2
1333 N/m2
When a perfect gas is expanded through an aperture of minute dimensions, the process is known as
Isothermal process
Adiabatic process
Free expansion process
Throttling process
The stress at which extension of the material takes place more quickly as compared to the increase in load is called
Elastic point of the material
Plastic point of the material
Breaking point of the material
Yielding point of the material
The behaviour of super-heated vapour is similar to that of
Perfect gas
Air
Steam
Ordinary gas
The volumetric or molar specific heat at constant pressure is the product of
Molecular mass of the gas and the specific heat at constant volume
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the gas constant
None of the above
The stress induced in a body, when suddenly loaded, is ________ the stress induced when the same load is applied gradually.
Equal to
One-half
Twice
Four times
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
Euler's formula holds good only for
Short columns
Long columns
Both short and long columns
Weak columns
The percentage reduction in area of a cast iron specimen during tensile test would be of the order of
More than 50 %
25-50 %
10-25 %
Negligible
The energy absorbed in a body, when it is strained within the elastic limits, is known as
Strain energy
Resilience
Proof resilience
Modulus of resilience
If the slenderness ratio for a column is 100, then it is said to be a _________ column.
Long
Medium
Short
None of these
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
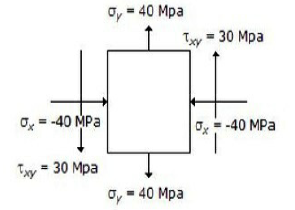
10 MPa
30 MPa
50 MPa
100 MPa
Which of the following statement is correct?
The heat and work are boundary phenomena
The heat and work represent the energy crossing the boundary of the system
The heat and work are path functions
All of the above
Which of the following materials is most elastic?
Rubber
Plastic
Brass
Steel
The efficiency of Joule cycle is
Greater than Carnot cycle
Less than Carnot cycle
Equal to Carnot cycle
None of these
For the same maximum pressure and temperature,
Otto cycle is more efficient than Diesel cycle
Diesel cycle is more efficient than Otto cycle
Dual cycle is more efficient than Otto and Diesel cycles
Dual cycle is less efficient than Otto and Diesel cycles
Which of the following process can be made reversible with the help of a regenerator?
Constant pressure process
Constant volume process
Constant pvn process
All of these
A thin cylindrical shell of diameter (D) and thickness (t) is subjected to an internal pressure (p). The ratio of longitudinal strain to volumetric strain is
(m - 1)/ (2m - 1)
(2m - 1)/ (m - 1)
(m - 2)/ (3m - 4)
(m - 2)/ (5m - 4)
If the rivets in adjacent rows are staggered and the outermost row has only one rivets, the arrangement of the rivets is called
Chain riveting
Zigzag riveting
Diamond riveting
Crisscross riveting
Flow stress corresponds to
Fluids in motion
Breaking point
Plastic deformation of solids
Rupture stress
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
The point of contra flexure is a point where
Shear force changes sign
Shear force is maximum
Bending moment changes sign
Bending moment is maximum
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
A molecule consisting of one atom is known as
Mono-atomic
Di-atomic
Tri-atomic
Poly-atomic
When both ends of a column are fixed, the effective length is
Its own length
Twice its length
Half its length
1/√2 × its length
The efficiency of Stirling cycle is __________ Carnot cycle.
Greater than
Less than
Equal to
None of these
