Which is the incorrect statement about Carnot cycle?
It is used as the alternate standard of comparison of all heat engines.
All the heat engines are based on Carnot cycle.
It provides concept of maximising work output between the two temperature limits.
All of the above
Correct Answer :
B. All the heat engines are based on Carnot cycle.
Related Questions
A process, in which the working substance neither receives nor gives out heat to its surroundings during its expansion or compression, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
All perfect gases change in volume by 1/273th of its original volume at 0°C for every 1°C change in temperature, when the pressure remains constant. This statement is called
Boyle's law
Charles' law
Gay-Lussac law
Joule's law
A boiler shell 200 cm diameter and plate thickness 1.5 cm is subjected to internal pressure of 1.5 MN/m, and then the hoop stress will be
30 MN/m²
50 MN/m²
100 MN/m²
200 MN/m²
The hard coke is obtained when carbonisation of coal is carried out at
300° to 500°C
500° to 700°C
700° to 900°C
900° to 1100°C
The shear force at the centre of a simply supported beam with a gradually varying load from zero at both ends to w per metre at the centre, is
Zero
wl/4
wl/2
wl²/2
A bar of copper and steel form a composite system, which is heated to a temperature of 40°C. The stress induced in the copper bar will be
Tensile
Compressive
Shear
Zero
The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
The stress induced in a body, when suddenly loaded, is ________ the stress induced when the same load is applied gradually.
Equal to
One-half
Twice
Four times
Which of the following has the highest calorific value?
Peat
Lignite
Bituminous coal
Anthracite coal
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
The general law for the expansion or compression of gases, is
pv = C
pv = m R T
pvn = C
pvγ = C
In the below figure, the stress corresponding to point D is
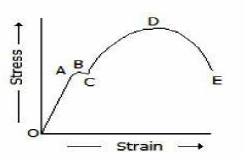
Yield point stress
Breaking stress
Ultimate stress
Elastic limit
Percentage reduction in area performing tensile test on cast iron may be of the order of
50 %
25 %
20 %
30 %
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
Impact strength of a material is an index of its
Toughness
Tensile strength
Capability of being cold worked
Hardness
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
Producer gas is obtained by
Partial combustion of coal, coke, anthracite coal or charcoal in a mixed air steam blast
Carbonisation of bituminous coal
Passing steam over incandescent coke
Passing air and a large amount of steam over waste coal at about 650°C
A process, in which the gas is heated or expanded in such a way that the product of its pressure and volume remains constant, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
A vertical column has two moments of inertia (i.e. Ixx and Iyy ). The column will tend to buckle in the direction of the
Axis of load
Perpendicular to the axis of load
Maximum moment of inertia
Minimum moment of inertia
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
The rivets are used for __________ fastenings.
Permanent
Temporary
Semi-permanent
None of these
In the below figure, curve D represents_________.
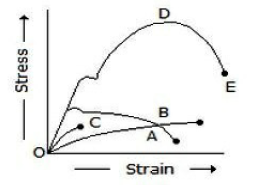
Mild steel
Cast iron
Concrete
Bone of these
According to Kelvin-Planck's statement of second law of thermodynamics,
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is possible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is impossible to construct a device which operates in a cyclic process and produces no effect other than the transfer of heat from a cold body to a hot body
None of the above
The materials which exhibit the same elastic properties in all directions are called
Homogeneous
Inelastic
Isotropic
Isentropic
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
The assumption made in Euler's column theory is that
The failure of column occurs due to buckling alone
The length of column is very large as compared to its cross-sectional dimensions
The column material obeys Hooke's law
All of the above
The efficiency of Joule cycle is
Greater than Carnot cycle
Less than Carnot cycle
Equal to Carnot cycle
None of these
In a tensile test on mild steel specimen, the breaking stress as compared to ultimate tensile stress is
More
Less
Same
More/less depending on composition
