Which of the following decreases with increase in pressure?
Melting point of ice
Melting point of wax
Boiling point of liquids
None of these
Correct Answer :
A. Melting point of ice
Related Questions
Which of the following processes cannot be made reversible even under ideal condition of operation?
Free expansion of a gas
Compression of air in a compressor
Expansion of steam in a turbine
All (A), (B) & (C)
The equation Tds = dE - PdV applies to
Single phase fluid of varying composition
Single phase fluid of constant composition
Open as well as closed systems
Both (B) and (C)
The heat capacities for the ideal gas state depend upon the
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
The entropy change in a reversible isothermal process, when an ideal gas expands to four times its initial volume is
R loge 4
R log10 4
Cv log10 4
Cv loge 4
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
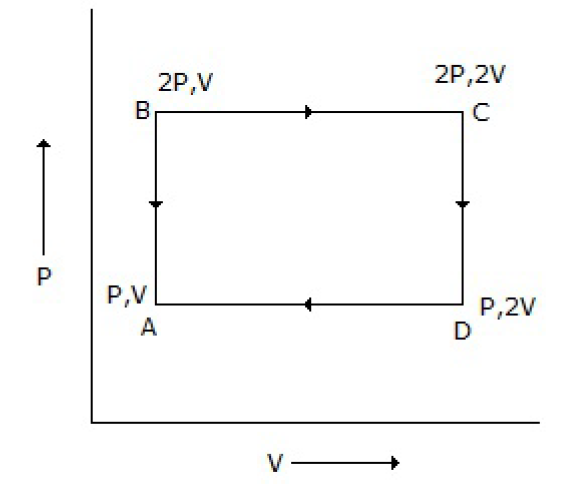
PV
2PV
PV/2
0
At a given temperature, the volume of a gas dissolved in a solvent __________ with increase in pressure.
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the gas
A system in which there is exchange of energy but not of mass, is called a/an __________ system.
Isolated
Open
Insulated
Closed
Entropy change in case of reversible adiabatic process is
Minimum
Zero
Maximum
Indeterminate
With increase in temperature, the internal energy of a substance
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the substance
At constant temperature and pressure, for one mole of a pure substance, the ratio of the free energy to the chemical potential is
Zero
One
Infinity
Negative
Entropy, which is a measure of the disorder of a system, is:
Independent of pressure
Independent of temperature
Zero at absolute zero temperature for a perfect crystalline substance
All (A), (B) & (C)
Free energy
Decreases in all spontaneous (or irreversible) processes
Change during a spontaneous process has a negative value
Remains unchanged in reversible processes carried at constant temperature and pressure
All (A), (B) and (C)
The necessary and sufficient condition for equilibrium between two phases is
The concentration of each component should be same in the two phases
The temperature of each phase should be same
The pressure should be same in the two phases
The chemical potential of each component should be same in the two phases
Throttling (Joule-Thomson effect) process is a constant __________ process.
Enthalpy
Entropy
Pressure
None of these
Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
(1/V) (∂V/∂T)P is the mathematical expression
Joule-Thomson co-efficient
Specific heat at constant pressure (Cp)
co-efficient of thermal expansion
Specific heat at constant volume (CV)
During a reversible isothermal expansion of an ideal gas, the entropy change is
+ve
0
-ve
∞
Tea kept in a thermos flask is vigorously shaken. If the tea is considered as a system, then its temperature will
Increase
Decrease
Remain unchanged
First fall and then rise
Pick out the wrong statement.
Cp of monatomic gases such as metallic vapor is about 5 kcal/kg.atom
The heat capacity of solid inorganic substance is exactly equal to the heat capacity of the substance in the molten state
There is an increase in entropy, when a spontaneous change occurs in an isolated system
At absolute zero temperature, the heat capacity for many pure crystalline substances is zero
Which of the following is not an intensive property?
Chemical potential
Surface tension
Heat capacity
None of these
Pick out the extensive property out of the following.
Surface tension
Free energy
Specific heat
Refractive index
Two substances are in equilibrium in a reversible chemical reaction. If the concentration of each substance is doubled, then the value of the equilibrium constant will be
Same
Doubled
Halved
One fourth of its original value
When a system in equilibrium is subjected to a change in temperature, pressure or concentration, the equilibrium is displaced in a direction which tends to undo the effect of the change. This is called the
Le-Chatelier principle
Kopp's rule
Law of corresponding state
Arrhenius hypothesis
4 kg moles of an ideal gas expands in vacuum spontaneously. The work done is
4 J
∞
0
8 J
A refrigerator may be termed as a
Heat pump
Heat engine
Carnot engine
None of these
Sound waves propagation in air exemplifies an __________ process.
Adiabatic
Isothermal
Isometric
None of these
Pick out the wrong statement.
A closed system does not permit exchange of mass with its surroundings but may permit exchange of energy.
An open system permits exchange of both mass and energy with its surroundings
The term microstate is used to characterise an individual, whereas macro-state is used to designate a group of micro-states with common characteristics
None of the above
__________ law of thermodynamics ascertains the direction of a particular spontaneous process.
Zeroth
First
Second
Third
A change in state involving a decrease in entropy can be spontaneous, only if
It is exothermic
It is isenthalpic
It takes place isothermally
It takes place at constant volume
The efficiency of a Carnot heat engine operating between absolute temperatures T1 and T2 (when, T1 > T2) is given by (T1 - T2)/T1. The co-efficient of performance (C.O.P.) of a Carnot heat pump operating between T1 and T2 is given by
T1/(T1-T2)
T2/(T1-T2)
T1/T2
T2/R1
