Which of the following has the highest calorific value?
Peat
Lignite
Bituminous coal
Anthracite coal
Correct Answer :
D. Anthracite coal
Related Questions
In open cycle gas turbine plants
The indirect heat exchanger and cooler is avoided
Direct combustion system is used
A condenser is used
All of the above
A beam which is fixed at one end and free at the other is called
Simply supported beam
Fixed beam
Overhanging beam
Cantilever beam
The value of 1 mm of Hg is equal to
1.333 N/m2
13.33 N/m2
133.3 N/m2
1333 N/m2
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
Strain is equal to (where l = Original length, and δl = Change in length)
l/δl
δl/l
l.δl
l + δl
One kg of carbon monoxide requires 4/7 kg of oxygen and produces
11/3 kg of carbon dioxide gas
7/3 kg of carbon monoxide gas
11/7 kg of carbon dioxide gas
8/3 kg of carbon monoxide gas
Those substances which have so far not been resolved by any means into other substances of simpler form are called
Elements
Compounds
Atoms
Molecules
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
The extension of a circular bar tapering uniformly from diameter d₁ at one end to diameter d₂ at the other end and subjected to an axial pull of P is given by
δl = 4PE/ πl²
δl = 4πld²/PE
δl = 4Pl/πEd₁d₂
δl = 4PlE/ πd₁d₂
A cylindrical section having no joint is known as
Joint less section
Homogeneous section
Perfect section
Seamless section
The most probable velocity of the gas molecules is given by
√(KT/m)
√(2KT/m)
√(3KT/m)
√(5KT/m)
The gas constant (R) is equal to the
Sum of two specific heats
Difference of two specific heats
Product of two specific heats
Ratio of two specific heats
According to kinetic theory of gases, the velocity of molecules __________ with the increase in temperature.
Remains constant
Increases
Decreases
None of these
For the beam shown in the below figure, the shear force diagram between A and B is
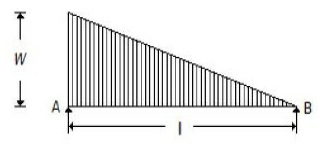
A horizontal line
A vertical line
An inclined line
A parabolic curve
The entropy __________ in an irreversible cyclic process.
Remains constant
Decreases
Increases
None of these
The value of modulus of elasticity for mild steel is of the order of
2.1 × 10⁵ kg/cm²
2.1 × 10⁶ kg/cm²
2.1 × 10⁷ kg/cm²
0.1 × 10⁶ kg/cm²
The processes occuring in open system which permit the transfer of mass to and from the system, are known as
Flow processes
Non-flow processes
Adiabatic processes
None of these
The heat absorbed or rejected by the working substance is given by (where ds = Increase or decrease of entropy, T = Absolute temperature, and dQ = Heat absorbed or rejected)
δQ = T.ds
δQ = T/ds
dQ = ds/T
None of these
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
One kg of ethylene (C2H4) requires 2 kg of oxygen and produces 22/7 kg of carbon dioxide and __________ kg of water or steam.
9/7
11/7
7/4
1/4
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
Petrol is distilled at
65° to 220°C
220° to 345°C
345° to 470°C
470° to 550°C
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
The specific heat at constant volume is
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant pressure
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
The amount of heat required to raise the temperature of 1 kg of water through one degree
Any one of the above
Which of the following is a reversible non-flow process?
Isochoric process
Isobaric process
Hyperbolic process
All of these
The efficiency of Ericsson cycle is __________ Carnot cycle.
Greater than
Less than
Equal to
None of these
High air-fuel ratio in gas turbines
Increases power output
Improves thermal efficiency
Reduces exhaust temperature
Do not damage turbine blades
Modulus of rigidity may be defined as the ratio of
Linear stress to lateral strain
Lateral strain to linear strain
Linear stress to linear strain
Shear stress to shear strain
One kg of carbon monoxide requires _______kg of oxygen to produce 11/7 kg of carbon dioxide gas.
11/7
9/7
4/7
All of the above
Which of the following statement is incorrect?
The liquid fuels consist of hydrocarbons.
The liquid fuels have higher calorific value than solid fuels.
The solid fuels have higher calorific value than liquid fuels.
A good fuel should have low ignition point.
