Which of the following is correct?
Absolute pressure = Gauge pressure + Atmospheric pressure
Gauge pressure = Absolute pressure + Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Correct Answer :
A. Absolute pressure = Gauge pressure + Atmospheric pressure
Related Questions
The entropy of water at 0°C is assumed to be
1
0
-1
10
The calorific value of gaseous fuel is expressed in
kJ
kJ/kg
kJ/m2
kJ/m3
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
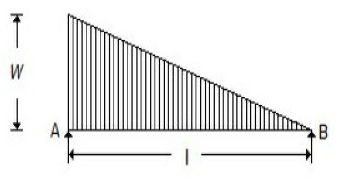
wl/6
wl/3
wl
2wl/3
The efficiency and work ratio of a simple gas turbine cycle are
Very low
Low
High
Very high
The specific heat at constant volume is
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant pressure
The amount of heat required to raise the temperature of unit mass of gas through one degree, at constant volume
The amount of heat required to raise the temperature of 1 kg of water through one degree
Any one of the above
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
Hooke's law holds good up to
Yield point
Limit of proportionality
Breaking point
Elastic limit
If Kh is the torque resisting capacity of a hollow shaft and Ks is that of a solid shaft, of the same material, length and weight. Then,
Kh > Ks
Kh < Ks
Kh = Ks
None of these
The property of a material by virtue of which a body returns to its original, shape after removal of the load is called
Plasticity
Elasticity
Ductility
Malleability
The thickness of a thin cylindrical shell with hemispherical ends is __________ that of spherical ends.
Equal to
More than
Less than
None of these
The processes occuring in open system which permit the transfer of mass to and from the system, are known as
Flow processes
Non-flow processes
Adiabatic processes
None of these
Which of the following cycles is not a reversible cycle?
Carnot
Ericsson
Stirling
None of the above
The maximum bending moment for the beam shown in the below figure, is
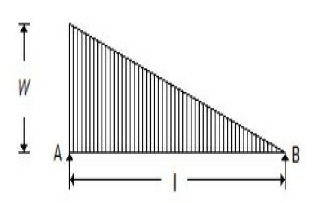
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
The compression ratio for petrol engines is
3 to 6
5 to 8
10 to 20
15 to 30
Those substances which have so far not been resolved by any means into other substances of simpler form are called
Elements
Compounds
Atoms
Molecules
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
When a body is subjected to two equal and opposite pushes, as a result of which the body tends to reduce its length, then
The stress and strain induced is compressive
The stress and strain induced is tensile
Both A and B is correct
None of these
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
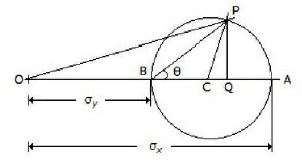
OC
OP
OQ
PQ
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
The compression ratio for petrol engines is
3 to 6
5 to 8
15 to 20
20 to 30
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
A cantilever beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported at its ends
Supported on more than two supports
The lower layer of the beam as shown in the below figure, will be

In tension
In compression
Neither in tension nor in compression
None of these
A process, in which the gas is heated or expanded in such a way that the product of its pressure and volume remains constant, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
A coil is cut into two halves, the stiffness of cut coil will be
Double
Half
Same
None of these
The compression ratio is the ratio of
Swept volume to total volume
Total volume to swept volume
Swept volume to clearance volume
Total volume to clearance volume
The gas turbine cycle with regenerator improves
Thermal efficiency
Work ratio
Avoids pollution
None of these
The pull required to tear off the plate per pitch length is (where p = Pitch of rivets, t = Thickness of plates, and σt, τ and σc = Permissible tensile, shearing and crushing stresses respectively)
(p - 2d) t × σc
(p - d) t × τ
(p - d) t × σt
(2p - d) t × σt
Which of the following gas has a minimum molecular mass?
Oxygen
Nitrogen
Hydrogen
Methane
The rivets are used for __________ fastenings.
Permanent
Temporary
Semi-permanent
None of these
