Which of the following is Virial equation of state?
(p + a/V2)(V - b) = nRT
PV = nRT
PV = A + B/V + C/V2 + D/V3 + ...
None of these
Correct Answer :
C. PV = A + B/V + C/V2 + D/V3 + ...
Related Questions
As the time is passing, entropy of the universe
Is increasing
Is decreasing
Remain constant
Data insufficient, can't be predicted
Pick out the correct statement.
A real gas on expansion in vacuum gets heated up
An ideal gas on expansion in vacuum gets cooled
An ideal gas on expansion in vacuum gets heated up
A real gas on expansion in vacuum cools down whereas ideal gas remains unaffected
The main feature of Carnot refrigeration cycle is that, it
Does not need the addition of external work for its functioning
Transfers heat from high temperature to low temperature
Accomplishes the reverse effect of the heat engine
None of these
Pick out the wrong statement.
A closed system does not permit exchange of mass with its surroundings but may permit exchange of energy.
An open system permits exchange of both mass and energy with its surroundings
The term microstate is used to characterise an individual, whereas macro-state is used to designate a group of micro-states with common characteristics
None of the above
Compressibility factor-reduced pressure plot on reduced co-ordinates facilitates
Use of only one graph for all gases
Covering of wide range
Easier plotting
More accurate plotting
Critical compressibility factor for all substances
Are more or less constant (vary from 0.2 to 0.3)
Vary as square of the absolute temperature
Vary as square of the absolute pressure
None of these
An isentropic process is carried out at constant
Volume
Pressure
Temperature
All (A), (B) and (C)
The point at which all the three (solid, liquid and gas) phases co-exist, is known as the __________ point.
Freezing
Triple
Boiling
Boyle
Refrigerants commonly used for domestic refrigerators are
Ethyl chloride or methyl chloride
Freon-12
Propane
NH3 or CO2
The unit of fugacity is the same as that of the
Pressure
Temperature
Volume
Molar concentration
The temperature at the eutectic point of the system is the __________ temperature that can be attained in the system.
Lowest
Highest
Average
None of these
Which of the following is not a reversible process?
Expansion of an ideal gas against constant pressure
Atmospheric pressure vaporisation of water at 100°C
Solution of NaCl in water at 50°C
None of these
4 kg moles of an ideal gas expands in vacuum spontaneously. The work done is
4 J
∞
0
8 J
Free energy change of mixing two liquid substances is a function of the
Concentration of the constituents only
Quantities of the constituents only
Temperature only
All (A), (B) and (C)
Change of heat content when one mole of compound is burnt in oxygen at constant pressure is called the
Calorific value
Heat of reaction
Heat of combustion
Heat of formation
The energy of activation of exothermic reaction is
Zero
Negative
Very large compared to that for endothermic reaction
Not possible to predict
The unit of equilibrium constant of a chemical reaction is the same as that of
Molar concentration
Temperature
Internal energy
None of these
A refrigerator works on the principle of __________ law of thermodynamics.
Zeroth
First
Second
Third
Pick out the wrong statement.
The conversion for a gas phase reaction increases with decrease in pressure, if there is an increase in volume accompanying the reaction
With increase in temperature, the equilibrium constant increases for an exothermic reaction
The equilibrium constant of a reaction depends upon temperature only
The conversion for a gas phase reaction increases with increase in pressure, if there is a decrease in volume accompanying the reaction
In case of a close thermodynamic system, there is __________ across the boundaries.
No heat and mass transfer
No mass transfer but heat transfer
Mass and energy transfer
None of these
Entropy is a measure of the __________ of a system.
Disorder
Orderly behaviour
Temperature changes only
None of these
The change in __________ is equal to the reversible work for compression in steady state flow process under isothermal condition.
Internal energy
Enthalpy
Gibbs free energy
Helmholtz free energy
An irreversible process
Is the analog of linear frictionless motion in machines
Is an idealised visualisation of behaviour of a system
Yields the maximum amount of work
Yields an amount of work less than that of a reversible process
__________ increases with increase in pressure.
The melting point of wax
The boiling point of a liquid
Both (A) and (B)
Neither (A) nor (B)
For a multi-component system, the term chemical potential is equivalent to the
Molal concentration difference
Molar free energy
Partial molar free energy
Molar free energy change
Consider the process A & B shown in the figure given below: In this case, it is possible that
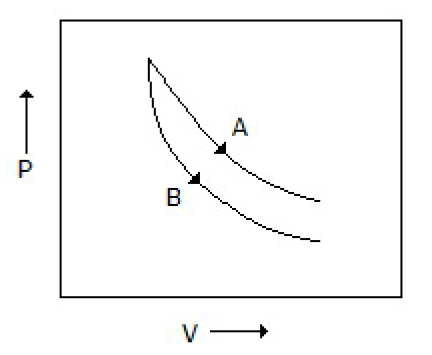
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
COP of a refrigerator drawing 1 kW of power per ton of refrigeration is about
0.5
3.5
4.5
8.5
In an ideal refrigeration cycle, the change in internal energy of the fluid is
+ve
-ve
0
Either of the above three; depends on the nature of refrigerant
At absolute zero temperature, the __________ of the gas is zero.
Pressure
Volume
Mass
None of these
A gas performs the maximum work, when it expands
Non-uniformly
Adiabatically
Isobarically
Isothermally
