Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
Correct Answer :
B. It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
Related Questions
The ratio of specific heat at constant pressure (cp) and specific heat at constant volume (cv) is always __________ one.
Equal to
Less than
Greater than
None of these
The heat and work are mutually convertible. This statement is called __________ law of thermodynamics.
Zeroth
First
Second
Third
In order to prevent crushing of masonry at the base of the dam, the maximum stress should be __________ the permissible stress of the soil.
Equal to
Less than
More than
None of these
The air standard efficiency of an Otto cycle is given by (where r = Compression ratio, and γ = Ratio of specific heats)
1 - rγ - 1
1 + rγ - 1
1 - (1/ rγ - 1)
1 + (1/ rγ - 1)
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
The molecular mass expressed in gram (i.e. 1 g - mole) of all gases, at N. T. P., occupies a volume of
0.224 litres
2.24 litres
22.4 litres
224 litres
Which of the following statement is correct according to Clausis statement of second law of thermodynamics?
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature.
It is impossible to transfer heat from a body at a lower temperature to a body at a higher temperature, without the aid of an external source.
It is possible to transfer heat from a body at a lower temperature to a body at a higher temperature by using refrigeration cycle.
None of the above
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
When a rectangular beam is loaded transversely, the maximum compressive stress is developed on the
Top layer
Bottom layer
Neutral axis
Every cross-section
In case of an under-reinforced beam, the depth of actual neutral axis is __________ that of the critical neutral axis.
Same as
Less than
Greater than
None of these
When two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This statement is called
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kelvin Planck's law
The strain energy stored in a solid circular shaft in torsion, subjected to shear stress (τ), is: (Where, G = Modulus of rigidity for the shaft material)
τ²/ 2G × Volume of shaft
τ/ 2G × Volume of shaft
τ²/ 4G × Volume of shaft
τ/ 4G × Volume of shaft
The bending equation is
M/I = σ/y = E/R
T/J = τ/R = Cθ/l
M/R = T/J = Cθ/l
T/l= τ/J = R/Cθ
Coke is produced
When coal is first dried and then crushed to a fine powder by pulverising machine
From the finely ground coal by moulding under pressure with or without a binding material
When coal is strongly heated continuously for 42 to 48 hours in the absence of air in a closed vessel
By heating wood with a limited supply of air to a temperature not less than 280°C
For riveting, the size of hole drilled in plates is __________ shank diameter of rivet.
Equal to
Less than
Greater than
None of these
A definite area or a space where some thermodynamic process takes place is known as
Thermodynamic system
Thermodynamic cycle
Thermodynamic process
Thermodynamic law
The radius of the Mohrs circle in the given figure is equal to
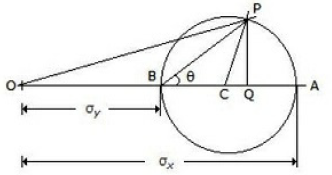
Sum of two principal stresses
Difference of two principal stresses
Half the sum of two principal stresses
Half the difference of two principal stresses
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
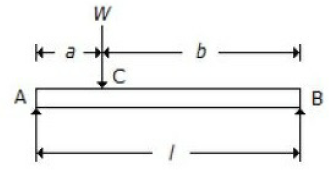
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
Diesel cycle consists of __________ processes.
Two constant volume and two isentropic
Two constant pressure and two isentropic
Two constant volume and two isothermal
One constant pressure, one constant volume and two isentropic
The __________ states that change of internal energy of a perfect gas is directly proportional to the change of temperature.
Boyle's law
Charle's law
Gay-Lussac law
Joule's law
The isothermal and adiabatic processes are regarded as
Reversible process
Irreversible process
Reversible or irreversible process
None of these
One molecule of oxygen consists of __________ atoms of oxygen.
2
4
8
16
The volumetric or molar specific heat at constant pressure is the product of
Molecular mass of the gas and the specific heat at constant volume
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the gas constant
None of the above
The bending stress in a beam is _________ section modulus.
Inversely proportional to two times
Directly proportional to
Inversely proportional to
None of these
A close cycle gas turbine gives __________ efficiency as compared to an open cycle gas turbine.
Same
Lower
Higher
None of these
The efficiency and work ratio of a simple gas turbine cycle are
Low
Very low
High
Very high
The maximum bending moment for the beam shown in the below figure, is
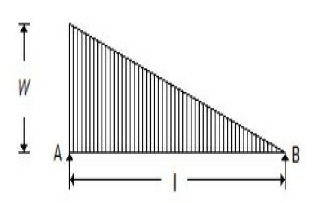
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
In an extensive property of a thermodynamic system
Extensive heat is transferred
Extensive work is done
Extensive energy is utilised
None of these
If the depth is kept constant for a beam of uniform strength, then its width will vary in proportional to
Bending moment (i.e. M)
Bending moment² (i.e. M²)
Bending moment³ (i.e. M³)
Bending moment⁴ (i.e. M⁴)
