With increase in compression ratio, the efficiency of the otto engine
Increases
Decreases
Remain constant
Increases linearly
Correct Answer :
A. Increases
Related Questions
The absolute entropy for all crystalline substances at absolute zero temperature is
Zero
Negative
More than zero
Indeterminate
Pick out the wrong statement.
At constant pressure, solubility of a gas in a liquid diminishes with rise in temperature
Normally, the gases which are easily liquefied are more soluble in common solvents
The gases which are capable of forming ions in aqueous solution are much more soluble in water than in other solvents
At constant pressure, solubility of a gas in a liquid increases with rise in temperature
The internal energy of an ideal gas does not change in a reversible __________ process.
Isothermal
Adiabatic
Isobaric
Isometric
The chemical potential of a component (μi) of a phase is the amount by which its capacity for doing all work, barring work of expansion is increased per unit amount of substance added for an infinitesimal addition at constant temperature and pressure. It is given by
(∂E/∂ni)S, v, nj
(∂G/∂ni)T, P, nj = (∂A/∂ni) T, v, nj
(∂H/∂ni)S, P, nj
All (A), (B) and (C)
Gibbs-Helmholtz equation is
ΔF = ΔH + T [∂(ΔF)/∂T]P
ΔF = ΔH - TΔT
d(E - TS) T, V < 0
dP/dT = ΔHvap/T.ΔVvap
Refrigeration cycle
Violates second law of thermodynamics
Involves transfer of heat from low temperature to high temperature
Both (A) and (B)
Neither (A) nor (B)
Pick out the wrong statement pertaining to the decomposition of PCl5 represented by, PCl5 PCl3 + Cl2.Degree of dissociation of PCl5 will
Decrease on addition of Cl2
Increase on addition of an inert gas at constant pressure
Decrease on increasing the pressure of the system
None of these
Enthalpy 'H' is defined as
H = E - PV
H = F - TS
H - E = PV
None of these
Gibbs free energy (G) is represented by, G = H - TS, whereas Helmholtz free energy, (A) is given by, A = E - TS. Which of the following is the Gibbs-Helmholtz equation?
[∂(G/T)/∂T] = - (H/T2)
[∂(A/T)/∂T]V = - E/T2
Both (A) and (B)
Neither (A) nor (B)
Sublimation temperature of dry ice (solid CO2) is __________ °C.
-273
0
-78
5
Specific __________ does not change during phase change at constant temperature and pressure.
Entropy
Gibbs energy
Internal energy
Enthalpy
The necessary condition for phase equilibrium in a multiphase system of N components is that the
Chemical potentials of a given component should be equal in all phases
Chemical potentials of all components should be same in a particular phase
Sum of the chemical potentials of any given component in all the phases should be the same
None of these
When a gas is subjected to adiabatic expansion, it gets cooled due to
Decrease in velocity
Decrease in temperature
Decrease in kinetic energy
Energy spent in doing work
When liquid and vapour phases of one component system are in equilibrium (at a given temperature and pressure), the molar free energy is
More in vapour phase
More in liquid phase
Same in both the phases
Replaced by chemical potential which is more in vapour phase
A domestic refrigerator has a/an __________ cooled condenser.
Water
Air
Evaporative
Gas
Variation of equilibrium pressure with temperature for any two phases of a given substances is given by the __________ equation.
Gibbs-Duhem
Maxwell's
Clapeyron
None of these
What happens in a reversible adiabatic expansion process?
Heating takes place
Cooling takes place
Pressure is constant
Temperature is constant
The efficiency of a Carnot heat engine operating between absolute temperatures T1 and T2 (when, T1 > T2) is given by (T1 - T2)/T1. The co-efficient of performance (C.O.P.) of a Carnot heat pump operating between T1 and T2 is given by
T1/(T1-T2)
T2/(T1-T2)
T1/T2
T2/R1
Equation which relates pressure, volume and temperature of a gas is called the
Equation of state
Gibbs Duhem equation
Ideal gas equation
None of these
The kinetic energy of gas molecule is zero at
0°C
273°C
100°C
-273°C
A gas has a volume of 27.3 c.c. at 0°C. Its volume at 10°C (if pressure remains unchanged) will be __________ c.c.
2.73
28.3
273
283
The activity of an ideal gas is numerically __________ its pressure.
More than
Less than
Equal to
Data insufficient, can't be predicted
Gibbs-Duhem equation
States that n1dμ1 + n2dμ2 + ....njdμj = 0, for a system of definite composition at constant temperature and pressure
Applies only to binary systems
Finds no application in gas-liquid equilibria involved in distillation
None of these
Enthalpy changes over a constant pressure path are always zero for __________ gas.
Any
A perfect
An easily liquefiable
A real
For organic compounds, group contribution method can be used for the estimation of
Critical properties
Specific gravity
Specific volume
Thermal conductivity
Ideal refrigeration cycle is
Same as Carnot cycle
Same as reverse Carnot cycle
Dependent on the refrigerant's properties
The least efficient of all refrigeration processes
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
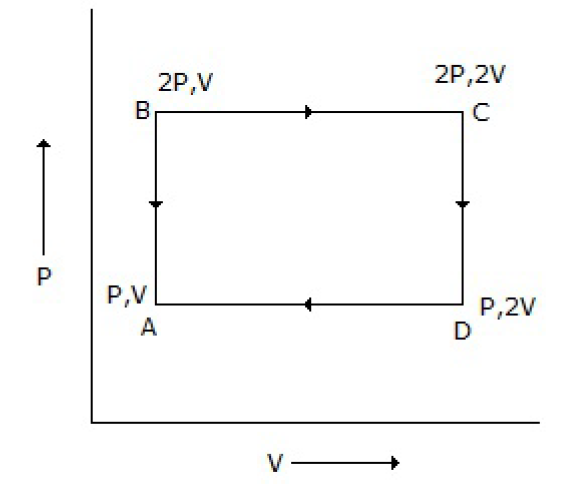
PV
2PV
PV/2
0
A refrigeration cycle is the same as a __________ cycle,
Turbine
Heat engine
Reversed heat engine
None of these
A solid metallic block weighing 5 kg has an initial temperature of 500°C. 40 kg of water initially at 25°C is contained in a perfectly insulated tank. The metallic block is brought into contact with water. Both of them come to equilibrium. Specific heat of block material is 0.4 kJ.kg-1. K-1. Ignoring the effect of expansion and contraction and also the heat capacity to tank, the total entropy change in kJ.kg-1, K-1 is
-1.87
0
1.26
3.91
Joule-Thomson co-efficient which is defined as, η = (∂T/∂P)H = 1/Cp (∂H/∂T)P, changes sign at a temperature known as inversion temperature. The value of Joule-Thomson co-efficient at inversion temperature is
0
∞
+ve
-ve
