Youngs modulus of a wire is defined as the stress which will increase the length of wire compared to its original length by
Half
Same amount
Double
One-fourth
Correct Answer :
B. Same amount
Related Questions
When a system changes its state from one equilibrium state to another equilibrium state, then the path of successive states through which the system has passed, is known as
Thermodynamic law
Thermodynamic process
Thermodynamic cycle
None of these
A cycle consisting of two constant pressure and two isentropic processes is known as
Carnot cycle
Stirling cycle
Otto cycle
None of these
Slenderness of a column is zero when
Ends are firmly fixed
Column is supported on all sides throughout the length
Length is equal to radius of gyration
Length is twice the radius of gyration
The gas turbine cycle with regenerator improves
Thermal efficiency
Work ratio
Avoids pollution
None of these
Stirling cycle consists of
Two constant volume and two isentropic processes
Two constant volume and two isothermal processes
Two constant pressure and two isothermal processes
One constant volume, one constant pressure and two isentropic processes
The principal constituents of a fuel are
Carbon and hydrogen
Oxygen and hydrogen
Sulphur and oxygen
Sulphur and hydrogen
Euler's formula holds good only for
Short columns
Long columns
Both short and long columns
Weak columns
Which of the following statement is incorrect?
The liquid fuels have higher calorific value than solid fuels
The solid fuels have higher calorific value than liquid fuels
A good fuel should have low ignition point
The liquid fuels consist of hydrocarbons
The following cycle is used for air craft refrigeration
Brayton cycle
Joule cycle
Carnot cycle
Reversed Brayton cycle
Carnot cycle has maximum efficiency for
Petrol engine
Diesel engine
Reversible engine
Irreversible engine
For the same maximum pressure and temperature,
Otto cycle is more efficient than Diesel cycle
Diesel cycle is more efficient than Otto cycle
Dual cycle is more efficient than Otto and Diesel cycles
Dual cycle is less efficient than Otto and Diesel cycles
The amount of heat required to raise the temperature of __________ water through one degree is called kilojoules.
1 g
10 g
100 g
1000 g
A path 1-2-3 is given. A system absorbs 100 kJ as heat and does 60 kJ of work while along the path 1-4-3, it does 20 kJ of work. The heat absorbed during the cycle 1-4-3 is 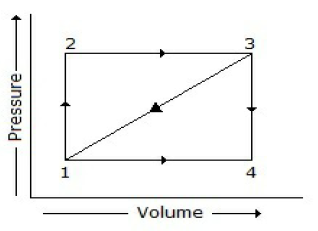
-140 kJ
-80 kJ
-40 kJ
+60 kJ
Strain is defined as the ratio of
Change in volume to original volume
Change in length to original length
Change in cross-sectional area to original cross-sectional area
Any one of the above
The absolute zero pressure will be
When molecular momentum of the system becomes zero
At sea level
At the temperature of - 273 K
At the centre of the earth
The area under the temperature-entropy curve (T - s curve) of any thermodynamic process represents
Heat absorbed
Heat rejected
Either (A) or (B)
None of these
Which is the incorrect statement about Carnot cycle?
It is used as the alternate standard of comparison of all heat engines.
All the heat engines are based on Carnot cycle.
It provides concept of maximising work output between the two temperature limits.
All of the above
For same compression ratio and for same heat added
Otto cycle is more efficient than Diesel cycle
Diesel cycle is more efficient than Otto cycle
Efficiency depends on other factors
Both Otto and Diesel cycles are equally efficient
According to Kelvin-Planck's statement of second law of thermodynamics,
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is possible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is impossible to construct a device which operates in a cyclic process and produces no effect other than the transfer of heat from a cold body to a hot body
None of the above
Impact strength of a material is an index of its
Toughness
Tensile strength
Capability of being cold worked
Hardness
The gas constant (R) is equal to the
Sum of two specific heats
Difference of two specific heats
Product of two specific heats
Ratio of two specific heats
The compression ratio for petrol engines is
3 to 6
5 to 8
15 to 20
20 to 30
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
The __________ states that change of internal energy of a perfect gas is directly proportional to the change of temperature.
Boyle's law
Charle's law
Gay-Lussac law
Joule's law
When a bar is cooled to - 5°C, it will develop
No stress
Shear stress
Tensile stress
Compressive stress
The compression ratio for petrol engines is
3 to 6
5 to 8
10 to 20
15 to 30
The variables which control the physical properties of a perfect gas are
Pressure exerted by the gas
Volume occupied by the gas
Temperature of the gas
All of these
The sum of internal energy (U) and the product of pressure and volume (p.v) is known as
Workdone
Entropy
Enthalpy
None of these
The limit of eccentricity for no tensile conditions for a column of circular section of diameter (D) is
d/4
d/8
d/12
d/16
The behaviour of a perfect gas, undergoing any change in the variables which control physical properties, is governed by
Boyle's law
Charles' law
Gay-Lussac law
All of these
