Atomic Structure 1000+ MCQ with answer for RRB JE
Thursday 9th of March 2023

Sharing is caring
1. rays are a stream of
A. He
B. electrons
C. positron
D. neutrons
Answer : B
A. He
B. electrons
C. positron
D. neutrons
Answer : B
2. The wavelength of an electron moving with a velocity
A.
B.
C.
D.
Answer : D
A.
B.
C.
D.
Answer : D
3. No two electrons in an atom can have the same values of all four quantum numbers according to
A. Hund's rules
B. Flemming rule
C. Pauli's exclusion principle
D. Bohr theory
Answer : C
A. Hund's rules
B. Flemming rule
C. Pauli's exclusion principle
D. Bohr theory
Answer : C
4. The angular momentum of the moving electron is equal to
A.
B.
C.
D.
Answer : D
A.
B.
C.
D.
Answer : D
5. The number of neutrons present in are
A. 28
B. 39
C. 20
D. 19
Answer : C
A. 28
B. 39
C. 20
D. 19
Answer : C
6. The corect set of quantum numbers for the impaired electron of fluorine atom is
A.
B.
C.
D.
Answer : B
A.
| n | l | m |
|---|---|---|
| 2 | 1 | 0 |
B.
| n | l | m |
|---|---|---|
| 2 | 1 | 1 |
C.
| n | l | m |
|---|---|---|
| 3 | 1 | 1 |
D.
| n | l | m |
|---|---|---|
| 3 | 0 | 0 |
Answer : B
7. The charge on an atom of is
A. +40
B. +27
C. +14
D. Zero
Answer : D
A. +40
B. +27
C. +14
D. Zero
Answer : D
8. Mass of positron is the same to that of
A. proton
B. meson
C. electron
D. neutron
Answer : C
A. proton
B. meson
C. electron
D. neutron
Answer : C
9. Among the following ions, which are has the highest paramagnetism?
A.
B.
C.
D.
Answer : B
A.
B.
C.
D.
Answer : B
10. Isotones have same
A. mass number
B. nuclear mass
C. no. of neutrons
D. no. of electrons
Answer : C
A. mass number
B. nuclear mass
C. no. of neutrons
D. no. of electrons
Answer : C
11. Which of the following atoms has a non-spherical outermost orbital.
A. H
B. Li
C. Be
D. B
Answer : D
A. H
B. Li
C. Be
D. B
Answer : D
12. The velocity of a photon is
A. dependent on its wavelength
B. dependent on its source
C. equal to cube of its amplitude
D. independent of its wavelength
Answer : D
A. dependent on its wavelength
B. dependent on its source
C. equal to cube of its amplitude
D. independent of its wavelength
Answer : D
13. Which of the following weighs the least?
A. 1 g of
B. 2 litres of at N.T.P.
C. molecules of
D. atoms of carbon
Answer : A
A. 1 g of
B. 2 litres of at N.T.P.
C. molecules of
D. atoms of carbon
Answer : A
14. Davisson and Germer gave an experimental evidence for
A. wave nature of electron
B. particle nature of electron
C. particle nature of light
D. wave nature of light
Answer : D
A. wave nature of electron
B. particle nature of electron
C. particle nature of light
D. wave nature of light
Answer : D
15. Electronic configuration of is
A.
B.
C.
D.
Answer : B
A.
B.
C.
D.
Answer : B
16. The interrelationship between matter and energy was given by
A. Rutherford
B. Joule
C. de Broglie
D. Einstein
Answer : D
A. Rutherford
B. Joule
C. de Broglie
D. Einstein
Answer : D
17. Charge on an electron was shown by
A. J. J. Thomson
B. Kirchoff
C. Ohm
D. M.Planck
Answer : A
A. J. J. Thomson
B. Kirchoff
C. Ohm
D. M.Planck
Answer : A
18. The ion that is isoelectronic with CO is
A.
B.
C.
D.
Answer : A
A.
B.
C.
D.
Answer : A
19. The maximum number of electrons that can be accommodated by an atom in g-sub-energy level are
A. 20
B. 25
C. 18
D. 12
Answer : C
A. 20
B. 25
C. 18
D. 12
Answer : C
20. The exchange of particles considered responsible for keeping the nucleons together are
A. meson
B. electron
C. positron
D. neutron
Answer : A
A. meson
B. electron
C. positron
D. neutron
Answer : A
21. Argon is isoelectronic with
A.
B.
C.
D.
Answer : B
A.
B.
C.
D.
Answer : B
22. The number of unpaired electrons in fluorine atoms is/are
A. 4
B. 2
C. 1
D. 0
Answer : C
A. 4
B. 2
C. 1
D. 0
Answer : C
23. A dipositive ion has in the K shell, 8 e~ s in the L shell and in the M shell. Atomic number of Z is
A. 19
B. 20
C. 16
D. 15
Answer : B
A. 19
B. 20
C. 16
D. 15
Answer : B
24. The atomic number of an element having the valence shell electronic configuration is
A. 35
B. 26
C. 23
D. 34
Answer : A
A. 35
B. 26
C. 23
D. 34
Answer : A
25. The wavelength of an electron
A. is equal to that of light
B. remains constant with velocity
C. decreases with an increasing velocity
D. increases with an decreasing velocity
Answer : C
A. is equal to that of light
B. remains constant with velocity
C. decreases with an increasing velocity
D. increases with an decreasing velocity
Answer : C
26. Which elements is represented by the following electronic configuration?
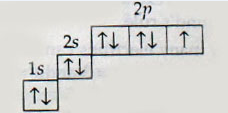
A. Nitrogen
B. Oxygen
C. Fluorine
D. Neon
Answer : C

A. Nitrogen
B. Oxygen
C. Fluorine
D. Neon
Answer : C
27. Frequency of the wave having wave number is
A.
B.
C.
D.
Answer : B
A.
B.
C.
D.
Answer : B
28. Cathode rays contain a stream of
A.
B.
C.
D.
Answer : D
A.
B.
C.
D.
Answer : D
29. The maximum number of electrons in a sub-energy level is equal to
A.
B.
C.
D.
Answer : B
A.
B.
C.
D.
Answer : B
30. The atomic structure of an atom of an element is Is . The element is
A. Cl-35
B. S-32
C. N-18
D. O-16
Answer : A
A. Cl-35
B. S-32
C. N-18
D. O-16
Answer : A
31. Bohr's concept of the orbit in an atom was contradicted by
A. de Broglie relationship
B. Uncertainty principle
C. Planck's hypothesis
D. Hund's rule
Answer : B
A. de Broglie relationship
B. Uncertainty principle
C. Planck's hypothesis
D. Hund's rule
Answer : B
32. The number of unpaired electrons in a chromic ion (atomic number 24) is
A. 6
B. 4
C. 3
D. 1
Answer : C
A. 6
B. 4
C. 3
D. 1
Answer : C
33. Wave number is defined as
A.
B.
C.
D.
Answer : D
A.
B.
C.
D.
Answer : D
34. Charge on fundamental particle neutrino is
A. 0
B. +1
C. -1
D. None of these
Answer : A
A. 0
B. +1
C. -1
D. None of these
Answer : A
35. In the nucleus of there are
A. 40 protons and 20 electrons
B. 20 protons and 40 electrons
C. 20 protons and 40 neutrons
D. 20 protons and 20 neutrons
Answer : D
A. 40 protons and 20 electrons
B. 20 protons and 40 electrons
C. 20 protons and 40 neutrons
D. 20 protons and 20 neutrons
Answer : D
36. The correct set of quantum numbers for the impaired electron of fluorine atom is
A.
B.
C.
D.
Answer : B
A.
| n | l | m |
| 2 | 1 | 0 |
B.
| n | l | m |
| 2 | 1 | 1 |
C.
| n | l | m |
| 3 | 1 | 1 |
D.
| n | l | m |
| 3 | 0 | 0 |
Answer : B
37. The orbitals with maximum number of possible orientation is:
A. s
B. d
C. f
D. p
Answer : C
A. s
B. d
C. f
D. p
Answer : C
38. In the case of atomic spectrum of hydrogen which series of lines lie in the visible region?
A. Balmer
B. Paschen
C. Pfund
D. None of these
Answer : B
A. Balmer
B. Paschen
C. Pfund
D. None of these
Answer : B
39. The fundamental particle that has least mass is
A. meson
B. alpha-particle
C. electron
D. neutron
Answer : D
A. meson
B. alpha-particle
C. electron
D. neutron
Answer : D
40. Which of the following has the highest mass?
A. Neutron
B. Alpha particle
C. Electron
D. Deuterium
Answer : B
A. Neutron
B. Alpha particle
C. Electron
D. Deuterium
Answer : B
41. The number of neutrons in the deuterium atom is
A. 16
B. 3
C. 1
D. 0
Answer : C
A. 16
B. 3
C. 1
D. 0
Answer : C
42. If value of h is taken as , the de-Broglie wavelength of a particle of mass having velocity is
A. 0.01 m
B. 2 nm
C. 0.1 nm
D.
Answer : C
A. 0.01 m
B. 2 nm
C. 0.1 nm
D.
Answer : C
43. For a given energy level the number of orbitals is equal to
A.
B.
C.
D.
Answer : A
A.
B.
C.
D.
Answer : A
44. In an atom of hydrogen, which of the following orbitais has the lowest energy for an electron present in it?
A. 3r
B. 2p
C. 4p
D. 2s
Answer : B
A. 3r
B. 2p
C. 4p
D. 2s
Answer : B
45. Isotopes have different
A. arrangement of electrons
B. no. of P and e'
C. no. of neutrons
D. no. of electrons
Answer : C
A. arrangement of electrons
B. no. of P and e'
C. no. of neutrons
D. no. of electrons
Answer : C
46. The number of unpaired electrons in the ground state of chromium is
A. 1
B. 6
C. 7
D. 2
Answer : B
A. 1
B. 6
C. 7
D. 2
Answer : B
47. The possible values of l for an s orbital are
A. -1, +1
B. 0 to 2
C. -2 and + 1
D. 0
Answer : D
A. -1, +1
B. 0 to 2
C. -2 and + 1
D. 0
Answer : D
48. The relationship between energy of a radiation and its frequency was given by
A. Planck
B. Rutherford
C. Einstein
D. Joule
Answer : C
A. Planck
B. Rutherford
C. Einstein
D. Joule
Answer : C
49. In Lyman series an electron jumps from higher energy level to
A. K energy level
B. M energy level
C. N energy level
D. L energy level
Answer : A
A. K energy level
B. M energy level
C. N energy level
D. L energy level
Answer : A
50. The size of nucleus is
A.
B.
C.
D.
Answer : C
A.
B.
C.
D.
Answer : C
Sharing is caring
Related Post
1000+ Puzzle MCQ for IBPS RRB [Solved]
IBPS RRB - Problems on Trains 1000+ MCQ [Solved] PDF Download
SSC CHSL - Current Affairs December 2022 1000+ MCQ [Solved] PDF Download
1000+ Linux OS Multiple Choice Question Answer [Solved]
UPSC CDS - Current Affairs January 2023 1000+ MCQ [Solved] PDF Download
RBI Grade B officer - Urinary Calculus Disease 1000+ MCQ [Solved] PDF Download